
Risk factors of cardiac complications in patients with end-stage renal disease undergoing maintenance peritoneal dialysis
Introduction
End-stage renal disease (ESRD) is the most severe stage of chronic kidney disease, and the risks of morbidity and mortality associated with ESRD have increased immensely worldwide (1), which, in turn, considerably increase the medical costs and global healthcare burden (2,3). More than 2 million patients have been receiving therapy for ESRD globally, and the prevalence of ESRD is expected to rise enormously in the future (1). Renal replacement therapy remains the only effective treatment when the disease worsens to ESRD. Moreover, dialysis is the primary therapy for ESRD as the scarcity of donors limits the possibility of kidney transplantation. However, severe complications are frequently encountered among patients despite undergoing maintenance dialysis, which is a reasonably effective treatment (4). The patients living with maintenance dialysis and cardiovascular complications have significantly increased morbidity and risk of mortality (5). The incidence of cardiovascular death in such patients is 5–25 times higher than that in the general population (6).
Peritoneal dialysis (PD) is an important therapeutic modality for patients with ESRD (7). A previous study indicated that the overall adjusted incidence of 1-year survival in patients on PD was 86.8%, and the incidence of 10-year survival was 11.3% (8). The decrease in the survival rate of patients on PD over time is robustly associated with multifaceted factors (7). Furthermore, cardiovascular disease (CVD) is the most frequent cause of death among patients on PD (9). Therefore, risk factors for CVD in this patient population need to be assessed and early interventions should be designed to improve the prognosis of patients with ESRD. This retrospective study was designed to identify the risk factors associated with cardiac complications in patients undergoing PD. We present the following article in accordance with the STROBE reporting checklist (available at https://apm.amegroups.com/article/view/10.21037/apm-21-2987/rc).
Methods
Study design and patient data collection
This was a single-centre, observational, retrospective study, in which all data were extracted from the medical records of patients with ESRD who underwent maintenance PD between January 2015 and June 2020 in the PD centre of Lixin People’s Hospital. A total of 232 patients were included in the study. The inclusion criteria were as follows: age ≥18 years, previously underwent maintenance PD (duration: >6 months), and diagnosis of ESRD. The exclusion criteria were as follows: refusal to follow-up, and missing follow-up data.
Data collection
Data on sociodemographic characteristics, concomitant diseases, treatment drugs, laboratory examination, and medical history were collected from the electronic medical records of patients, and all these data was before the data of their first PD. The following variables was analysed: age; sex; marital status (married/widowed or bachelor); educational level (under high school/high school or above); systolic and diastolic blood pressure; coronary artery disease; hypertension; diabetic nephropathy; diabetes mellitus; history of stroke; medication history of calcium channel blockers, diuretics, beta-blockers, calcium supplements, antiplatelet drugs, and insulin; and laboratory data of serum uric acid, chlorine, hemoglobin, alanine transaminase, aspartate aminotransferase, alkaline phosphatase, albumin, blood urea nitrogen, creatinine, β2-microglobulin, potassium, sodium, calcium, phosphate, fasting blood-glucose, triglyceride, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), C-reactive protein (CRP), creatine kinase, lactate dehydrogenase, creatine kinase isoenzyme, ferritin, D-dimer, total iron-binding capacity, transferrin, parathyroid hormone, folic acid, and erythrocyte and leukocyte counts. We also retrieved the data of Doppler echocardiography: left ventricular ejection fraction (LVEF), left atrial diameter, pulmonary arterial hypertension and checked for any findings of ST-segment depression in the electrocardiography data.
Endpoints and outcome assessment
The primary outcome variable was the occurrence of cardiac complications in the first 6 months of PD. Cardiac complications including acute heart failure, incidence of cardiac death, unstable angina, myocardial infarction, cardiovascular comorbidity, and cardiovascular hospitalisation were evaluated in the analysis.
Statistical analysis
Data are presented as number (%) or mean ± standard deviation. Continuous variables were evaluated using the t-test or Mann-Whitney U test, and categorical variables were evaluated using the χ2 or Fisher exact test for analysing the intergroup differences. Statistical significance was set at P<0.05. All variables were examined by univariate analysis, and variables with P<0.100 in the univariate analysis were included in a multivariate logistic regression model. Multivariate logistic regression analysis was performed to determine the independent risk factors that could precisely predict the cardiac complications.
The performance of the multivariate logistic regression model was evaluated using the receiver operating characteristic (ROC) curve. All statistical analyses were conducted using IBM SPSS 24.0 (IBM Corporation, Armonk, New York, USA) and R statistical software (version 4.0.5; http://www.Rproject.org).
Ethical statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Medical Ethics Committee of the Lixin People’s Hospital (approval No. LXH-2021T001) and individual consent for this retrospective analysis was waived.
Results
A total of 232 patients who fulfilled the inclusion criteria between January 2015 and June 2020 were enrolled (Figure S1). The included participants had an average age of 51.29±13.17 years, with female-to-male ratio of 87:145, and 119 patients had cardiac complications. The description of categorical variables in our study is shown in Table S1. In our study data, some variables have incomplete data, and the baseline characteristics of patients are shown in Table S2.
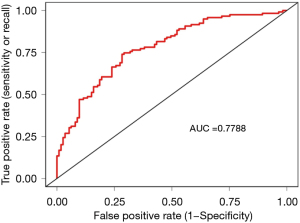
Data of variables with P<0.100 in the univariate analysis, including hemoglobin, albumin, serum calcium, creatine kinase, lactate dehydrogenase, creatine kinase isoenzyme, D-dimer, left atrial diameter, sex, coronary artery disease, hypertension, diuretics, antiplatelet drugs, diabetic nephropathy, diabetes mellitus, and use of insulin, were included in a multivariate logistic regression model (Table 1). We identified lactate dehydrogenase [odds ratio (OR), 1.002; 95% confidence interval (CI), 1.001–1.004; P=0.004], albumin (OR, 0.947; 95% CI, 0.914–0.982; P=0.003), and left atrial diameter (OR, 1.096; 95% CI, 1.037–1.159; P=0.001) as independent risk factors for cardiac complications (Table 2). We plotted the ROC curve to assess the performance of the multivariate logistic regression model and calculated the area under the ROC curve (AUROC) to obtain the AUROC score. The AUROC was 0.78 (Figure 1).
Table 1
| Variables | Total | No cardiac complications (N=113; 48.7%) | Cardiac complications (N=119; 51.3%) | P value |
|---|---|---|---|---|
| Hemoglobin (g/L) | 232 (100%) | 90.74±19.94 | 86.16±18.86 | 0.055 |
| Albumin (g/L) | 232 (100%) | 38.33±8.09 | 34.45±8.33 | <0.001 |
| Serum calcium (mmol/L) | 232 (100%) | 1.97±0.28 | 1.91±0.27 | 0.080 |
| Creatine Kinase (IU/L) | 232 (100%) | 190.21±233.53 | 269.84±262.08 | <0.001 |
| Lactate dehydrogenase (IU/L) | 232 (100%) | 262.92±136.76 | 408.51±377.47 | <0.001 |
| Creatine kinase isoenzyme (IU/L) | 232 (100%) | 14.49±8.18 | 20.50±33.84 | 0.019 |
| D-dimer (mg/L) | 232 (100%) | 0.55±0.83 | 0.85±1.29 | 0.010 |
| Left atrial diameter (mm) | 232 (100%) | 31.35±5.34 | 34.60±5.66 | <0.001 |
| Sex | 232 (100%) | 0.019 | ||
| Male | 62 (42.8%) | 83 (57.2%) | ||
| Female | 51 (58.6%) | 36 (41.4%) | ||
| Coronary artery disease | 232 (100%) | 0.031 | ||
| Yes | 4 (23.5%) | 13 (76.5%) | ||
| No | 109 (50.7%) | 106 (49.3%) | ||
| Hypertension | 232 (100%) | 0.023 | ||
| Yes | 99 (46.5%) | 114 (53.5%) | ||
| No | 14 (73.7%) | 5 (26.3%) | ||
| Diuretic | 232 (100%) | 0.074 | ||
| Yes | 26 (39.4%) | 40 (60.6%) | ||
| No | 87 (52.4%) | 79 (47.6%) | ||
| Antiplatelet drugs | 232 (100%) | 0.080 | ||
| Yes | 6 (30.0%) | 14 (70.0%) | ||
| No | 107 (50.5%) | 105 (49.5%) | ||
| Diabetic nephropathy | 232 (100%) | 0.002 | ||
| Yes | 8 (24.2%) | 25 (75.8%) | ||
| No | 105 (52.8%) | 94 (47.2%) | ||
| Diabetes mellitus | 232 (100%) | 0.002 | ||
| Yes | 11 (26.8%) | 30 (73.2%) | ||
| No | 102 (53.4%) | 89 (46.6%) | ||
| Use of insulin | 232 (100%) | 0.002 | ||
| Yes | 10 (25.6%) | 29 (74.4%) | ||
| No | 103 (53.4%) | 90 (46.6%) |
Data were N (%) or mean ± standard deviation. Continuous variables used Mann-Whitney U test and categorical variables used chi-squared test for comparing the baseline characteristics of patients with cardiac complications and without cardiac complications.
Table 2
| Variables | B | OR | 95% CI, lower | 95% CI, upper | P value |
|---|---|---|---|---|---|
| Lactate dehydrogenase | 0.002 | 1.002 | 1.001 | 1.004 | 0.004 |
| Albumin | −0.054 | 0.947 | 0.914 | 0.982 | 0.003 |
| Left atrial diameter | 0.092 | 1.096 | 1.037 | 1.159 | 0.001 |
B, partial regression coefficient; OR, odds ratio; CI, confidence interval.
Discussion
In our study, we focused on finding and assessing the independent risk factors for cardiac complications in PD patients. From the overall analysis, lactate dehydrogenase, albumin, and left atrial diameter were determined as the independent risk factors for cardiac complications in the PD population. In addition, the area under the curve score of the multivariate logistic regression model was 0.78. Our results may help clinicians to assess patients with ESRD on maintenance PD and prepare for their timely interventions, which would help improve their prognosis.
The level of serum albumin is an important marker to assess the status of nutritional in ESRD patients (10,11). Previous studies indicated that low serum albumin level was strongly associated with mortality and had a prognostic value for major adverse events (12-15). Therefore, albumin levels could contribute to risk profiling and identifying the relative cardiorenal factors in patients undergoing PD. Our results suggest that albumin is an independent risk factor for cardiac complications in patients undergoing PD, which is consistent with the findings of previous studies. The level of low serum albumin may be a consequence of ESRD and not a cause, but it was associated with the adverse outcomes of patients in PD.
Previous studies have reported that the majority of dialysis patients have left ventricular hypertrophy and dysfunction, which are critical cardiac complications that may occur in patients on PD (16,17). When the left atrial diameter of patients on PD was compared over time, a significant difference was noted (18). The left atrial diameter may precisely reflect the left ventricular diastolic function in patients undergoing PD (19). Likewise, in the present study, the left atrial diameter was found to be an independent risk factor for cardiac complications, which may provide a useful basis for clinical studies. Furthermore, lactate dehydrogenase was reported as a significant risk factor of cardiac complications. However, we believe that further high-quality studies are warranted to verify these findings.
Prediction models of cardiac risk factors have been frequently used (20-23), which has improved the clinical outcomes of patients. The prediction model for cardiovascular mortality can contribute to enact specific interventions (24); however, there is a lack of related study to determinate the risk factors of cardiac complications for patients on maintenance PD. In our study, we determined the risk factors of cardiac complications in patients undergoing PD. This may help provide early treatments and ameliorate the clinical outcomes of such patients. To identify the high-risk patients undergoing PD, cardiovascular risk stratification is a crucial step for their assessment; thus, early initiation of interventions and therapy can help decrease the cardiovascular morbidity and mortality (25).
The present study had some limitations. First, data on some biomarkers of laboratory tests, such as homocysteine and troponin, were lacking; moreover, the laboratory data were incomplete for some patients, potential confounders may be present in the data, which may have affected our findings. Second, the retrospective, observational nature of the study might have negatively affected its quality. Third, the sample size of this study was relatively small because the data were collected from a single hospital, which may be unable to perform regression model to capture potential risk factors. Lastly, the duration of follow-up was 6 months; hence, studies with long-term follow-up are warranted in future to verify our findings.
Acknowledgments
We thanks to Medical English Editing Service (https://www.editage.cn) for helping to check the wordings of our manuscript.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://apm.amegroups.com/article/view/10.21037/apm-21-2987/rc
Data Sharing Statement: Available at https://apm.amegroups.com/article/view/10.21037/apm-21-2987/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://apm.amegroups.com/article/view/10.21037/apm-21-2987/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Medical Ethics Committee of the Lixin People’s Hospital (approval No. LXH-2021T001) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Liyanage T, Ninomiya T, Jha V, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet 2015;385:1975-82. [Crossref] [PubMed]
- Eriksson JK, Neovius M, Jacobson SH, et al. Healthcare costs in chronic kidney disease and renal replacement therapy: a population-based cohort study in Sweden. BMJ Open 2016;6:e012062. [Crossref] [PubMed]
- Wyld ML, Lee CM, Zhuo X, et al. Cost to government and society of chronic kidney disease stage 1-5: a national cohort study. Intern Med J 2015;45:741-7. [Crossref] [PubMed]
- Robinson BM, Akizawa T, Jager KJ, et al. Factors affecting outcomes in patients reaching end-stage kidney disease worldwide: differences in access to renal replacement therapy, modality use, and haemodialysis practices. Lancet 2016;388:294-306. [Crossref] [PubMed]
- Prichard S. Cardiovascular risk in peritoneal dialysis. Contrib Nephrol 2003;82-90. [Crossref] [PubMed]
- Foley RN, Parfrey PS, Sarnak MJ. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am J Kidney Dis 1998;32:S112-9. [Crossref] [PubMed]
- Kendrick J, Teitelbaum I. Strategies for improving long-term survival in peritoneal dialysis patients. Clin J Am Soc Nephrol 2010;5:1123-31. [Crossref] [PubMed]
- Piraino B. Cardiovascular complications in peritoneal dialysis patients. Contrib Nephrol 2009;163:102-9. [Crossref] [PubMed]
- Krediet RT, Balafa O. Cardiovascular risk in the peritoneal dialysis patient. Nat Rev Nephrol 2010;6:451-60. [Crossref] [PubMed]
- Fouque D, Pelletier S, Mafra D, et al. Nutrition and chronic kidney disease. Kidney Int 2011;80:348-57. [Crossref] [PubMed]
- John B, Tan BK, Dainty S, et al. Plasma volume, albumin, and fluid status in peritoneal dialysis patients. Clin J Am Soc Nephrol 2010;5:1463-70. [Crossref] [PubMed]
- Mehrotra R, Duong U, Jiwakanon S, et al. Serum albumin as a predictor of mortality in peritoneal dialysis: comparisons with hemodialysis. Am J Kidney Dis 2011;58:418-28. [Crossref] [PubMed]
- Adequacy of dialysis and nutrition in continuous peritoneal dialysis: association with clinical outcomes. Canada-USA (CANUSA) Peritoneal Dialysis Study Group. J Am Soc Nephrol 1996;7:198-207. [Crossref] [PubMed]
- Blake PG, Flowerdew G, Blake RM, et al. Serum albumin in patients on continuous ambulatory peritoneal dialysis--predictors and correlations with outcomes. J Am Soc Nephrol 1993;3:1501-7. [Crossref] [PubMed]
- Spiegel DM, Anderson M, Campbell U, et al. Serum albumin: a marker for morbidity in peritoneal dialysis patients. Am J Kidney Dis 1993;21:26-30. [Crossref] [PubMed]
- Han SS, Cho GY, Park YS, et al. Predictive value of echocardiographic parameters for clinical events in patients starting hemodialysis. J Korean Med Sci 2015;30:44-53. [Crossref] [PubMed]
- Wang AY, Wang M, Woo J, et al. Inflammation, residual kidney function, and cardiac hypertrophy are interrelated and combine adversely to enhance mortality and cardiovascular death risk of peritoneal dialysis patients. J Am Soc Nephrol 2004;15:2186-94. [Crossref] [PubMed]
- Shi Q, Zhu J, Feng S, et al. Nonparallel Progression of Left Ventricular Structure and Function in Long-Term Peritoneal Dialysis Patients. Cardiorenal Med 2017;7:198-206. [Crossref] [PubMed]
- Ie EH, Vletter WB, ten Cate FJ, et al. Preload dependence of new Doppler techniques limits their utility for left ventricular diastolic function assessment in hemodialysis patients. J Am Soc Nephrol 2003;14:1858-62. [Crossref] [PubMed]
- Hijazi Z, Lindahl B, Oldgren J, et al. Repeated Measurements of Cardiac Biomarkers in Atrial Fibrillation and Validation of the ABC Stroke Score Over Time. J Am Heart Assoc 2017;6:004851. [Crossref] [PubMed]
- Fu Q, Wang XJ, Wang BS, et al. Cardiac risk prediction model for pregnant women with structural heart disease in Eastern China. Int J Gynaecol Obstet 2019;145:324-30. [Crossref] [PubMed]
- Gupta PK, Gupta H, Sundaram A, et al. Development and validation of a risk calculator for prediction of cardiac risk after surgery. Circulation 2011;124:381-7. [Crossref] [PubMed]
- Nakajima K, Nakata T, Yamada T, et al. A prediction model for 5-year cardiac mortality in patients with chronic heart failure using 123I-metaiodobenzylguanidine imaging. Eur J Nucl Med Mol Imaging 2014;41:1673-82. [Crossref] [PubMed]
- Xia X, Zhao C, Luo Q, et al. Nomogram for Predicting Cardiovascular Mortality in Incident Peritoneal Dialysis Patients: An Observational Study. Sci Rep 2017;7:13889. [Crossref] [PubMed]
- Mahmood U, Johnson DW, Fahim MA. Cardiac biomarkers in dialysis. AIMS Genet 2016;4:1-20. [Crossref] [PubMed]


