
The minimally invasive spinal surgery in the treatment of posterior edge separation
Introduction
Posterior edge separation of the vertebral body (also known as metaphysis of posterior margin of vertebral body) is an uncommon but non-rare orthopedic disease, in which the osteoid tissue located in the posterior edge of the affected vertebra compresses the central spinal canal or nerve root canal and then pain occurs in the lower back and/or sciatic nerve striding area. The disease was first reported by Skobowytsh-Okolot (1) in 1962, and its specific cause and pathogenesis have not been clarified. Posterior edge separation frequently occurs in young and middle-aged adults, and the lesions are mostly concentrated in the lower lumbar spine (the L3-S1 space accounts for 97%). When it comes to the diagnosis and treatment of this disease, due to the constant compression of the hard bone at the posterior edge of the vertebral body on the nerve root/spinal canal, especially when the compression has led to obvious changes in root symptoms of lower limbs (abnormal muscle strength and sensation et al.), surgical treatment has become a problem that needs to be considered by both doctors and patients. The traditional methods are posterior open surgery, based on the principle of complete decompression, such as fenestration, semi-laminectomy and total laminectomy, to name a few. In recent years, with the development of minimally invasive technology in spine surgery and the update of equipment, scholars have made a lot of attempts in minimally invasive treatment of lumbar posterior edge fracture, such as PELD, MISS-TLIF and Posterior Lumbar Interbody Fusion.
To discover the value of minimally invasive surgery in the treatment of posterior edge separation, we designed this retrospective study. From September 2015 to May 2019, 18 patients with posterior edge separation who underwent minimally invasive surgical treatment in the third affiliated hospital of Jinzhou Medical University were included in this study. All had received satisfactory clinical efficacy. We present the following article in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting checklist (available at http://dx.doi.org/10.21037/apm-20-928).
Methods
Patients’ characteristics
From September 2015 to May 2019, second department of orthopaedics in the third affiliated hospital of Jinzhou Medical University had treated a total of 18 patients (including 10 males and 8 females) with posterior edge separation, aged 19–69 years old (average age: 49.9 years old). Segmental distribution: 2 case of posterior lower margin of L2 vertebral body, 3 cases of posterior lower margin of L3 vertebral body, 8 cases of posterior lower margin of L4 vertebral body, and 5 cases of posterior lower margin of L5 vertebral body. The course of the disease ranges from 1 month to 9 years. Patients with lumbar instability, spondylolisthesis or large bony fragment, spinal canal stenosis were treated by MISS-TLIF.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Written informed consent for the experimental studies was obtained from the patients and all experiments were approved by the Institutional Ethics Committee of Third Affiliated Hospital of Jinzhou, Medical University.
Diagnostic criteria
The diagnosis of posterior edge vertebral detachment is generally not difficult, according to patient's symptoms, signs and related imaging data. The patients often complain of the pain of lumbar leg place, numb, send cool et al. Physical examination may show paravertebral radioactive tenderness, lower extremity paresthesia, abnormal muscle strength, and tendon reflex, while straight leg elevation test and straight leg elevation strengthening tests also could be positive. As for imaging, regular lateral and positive X-ray radiographs of the lumbar spine occasionally showed triangular bone segments separated from the posterior edge of the vertebral body; T2-weighted MRI revealed “curved” or “irregular” areas of low signal in the bone defect area at the posterior edge of the vertebral body. In particular, due to the unique advantages of bone structure, CT examination plays a key role in the diagnosis of the disease: the sagittal CT scan can detect the bone defect in the posterior superior/inferior margin of the vertebral body and the defect bone in the protruding vertebral canal, while the transverse scan can clearly show the detached vertebral body, the defect bone and the displacement into the vertebral canal.
Surgical methods
Lumbar instability Patients in group A (9 cases) were treated with PELD under local anesthesia, while patients in group B (9 cases) were treated with MISS-TLIF under general anesthesia. In both groups, the patients were prone at the translucency lumbar bridge, with slight knee flexion at the hip position, and extra soft pads were placed at the elbow and knee joints and the protruding part of the abdominal side of the body to prevent the compression of skin and other soft tissues from being compressed during the operation. The surgical plan was set according to the size and location of the detached bone mass and whether it is combined with lumbar instability/spondylolisthesis, articular process joint hyperplasia and cohesion, fibrous lateral recess stenosis and other degenerative changes.
Inclusion criteria for group A who were treated with PELD: CT and MRI of the lower lumbar spine showed isolated bone mass at the posterior edge of the vertebral body of a single segment, with small bone mass and no spinal stenosis or lumbar vertebral body instability. Inclusion criteria for group B: the results of imaging examination indicated that the detached bone mass was large and strongly adhered to the dural sac, which was accompanied by degenerative changes such as small joint hyperplasia and cohesion, lateral recess stenosis and lumbar body instability. MISS-TILIF surgery was then performed.
Postoperative management
Patients in the PELD group lay in bed for 24 hours and ambulated with the aid of hard waist circumference. After 4–6 weeks of surgery, nerve pulling exercises were performed to prevent nerve adhesion. And 6–8 weeks after the operation, lumbar dorsal muscle functional exercise was applied. As for the MISS-TLIF group, patients’ ambulation was allowed 4 days after surgery, and the incision suture was removed 15 days later.
Efficacy evaluation and statistical analysis
VAS scores and Oswestry disability index (ODI) index scores were obtained three times for all patients: before surgery, three days after surgery and at the last follow-up. Independent sample t-test was used to compare the difference in the corresponding time point effect between groups A and B before and after treatment. The paired t test was applied to compare the differences before and after treatment of each surgical procedure. P values <0.05 were considered statistically significant.
Results
The clinical characteristics of patients included in this study are listed in Table 1. Results of minimally invasive spinal surgery: in the PELD group, the average operation time was 71 min, the average incision length was 6.8 mm, the average intraoperative blood loss was 5 mL, and postoperative ambulation occurred by postoperative day one. In the MISS-TLIF group, the average operation time was 105min, the average incision length was 31.2 mm, the average intraoperative blood loss was 101ml, and postoperative ambulation occurred by postoperative day four. Neither group A nor group B experienced intraoperative dural sac and spinal nerve injury, and there were no postoperative complications such as delayed wound healing and infection. Postoperative follow-up was conducted from 3 months to 1 year, with an average duration of 9 months (1 person in PELD group was lost to follow-up). VAS score of patients in group A improved from 7.82±1.18 points preoperatively to 1.26±0.58 points (P<0.05); ODI score improved from 38.76±10.52 points to 10.02±6.45 points (P<0.05). While patients in group B who were treated with MISS-TLIF surgery, the VAS score improved from the preoperative average of 7.52±1.62 to 1.28±1.59 (P<0.05), ODI index improved from the preoperative average of 38.35±8.33 to 9.97±5.22 (P<0.05), and back pain function improved.
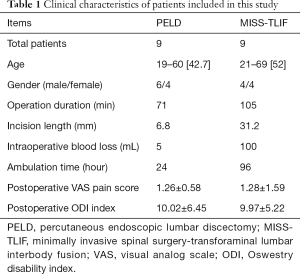
Full table
Typical cases
Medical records
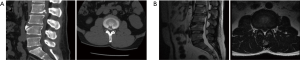
A 29-year-old male patient was admitted to the hospital with the chief complaint of “low back pain accompanied by right lower limb numbness for 3 years with aggravation for half a month”. Physical examination: 2 cm tenderness on the right side of the paraspinal space of lumbar 2/3 spinous process was positive; the anterolateral skin sensation on the right thigh is diminished; bilateral knee reflex and Achilles tendon reflex are normal; bilateral straight leg elevation test (−), femoral nerve traction test (+); primitive reflexes, but no pathological reflex, were elicited; preoperative visual analogue scale (VAS) score was 7; and as shown in Figure 1, preoperative CT and MRI of the lower lumbar vertebra indicated that the fractured bone at the posterior lower margin of L2 vertebral body protruded toward the central vertebral canal, and the dura sac was compressed.
Surgical procedure
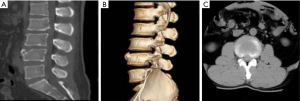
Preoperative positioning: the patient lies on the tummy in the permeable X-ray surgical bed. Determine the intraoperative operation path and needle orientation under G arm fluoroscopy, and duplication of preoperative marking from a permanent marker pen. The surfaces were sterilized by routine disinfection and draping, followed with local lidocaine anesthesia and No. 18 puncture needle was applied. As shown in Figure 2A, the needle trajectory angle was determined by spines on the attachment and the horizontal plane, both at an angle of 45 degrees. Under orthotopic fluoroscopy, the puncture tip was placed on the line of lumbar spinous process, while the lateral fluoroscopy tip was located on the line of the posterior edge of the lumbar 2/3 vertebral body, and local anesthesia was given consequently (Figure 2B). The guide wire was inserted, the puncture needle was removed, and the skin incision was made with a sharp blade with (approx. 7 mm long). The first stage guide rod, second, third and fourth stage catheter were placed in turn, and the articular process arthroplasty was performed. The catheter was removed and the work channel was placed. The position of the channel was determined under the perspective, and the position was satisfactory (Figure 2C). The imaging system was connected, and the radiofrequency electrode was used to stop bleeding and ablate the surrounding tissues of the nerve root. And bone mass of the posterior margin of the lumbar 2 vertebral body and the intervertebral disc tissue around the nerve root of the lumbar 3 were removed carefully and thoroughly. Annulus fibrosus and intervertebral disc tissues were ablated, and no obvious compression was observed, making sure of relaxation in lumbar nerve root 3. No significant active bleeding was observed in the operative field. At the end of the procedure, the operating system and working channel were removed, the incision was sutured, and the sterile dressing was applied to the surgical incision.
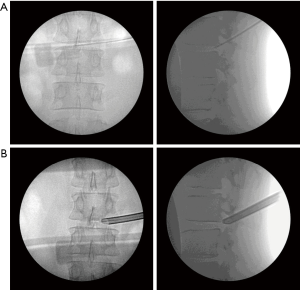
The patient experienced significant relief of pain immediately after surgery (VAS score 0). There were no complications such as nerve root injury, hematoma formation and dural sac tear during the procedure. Postoperative ambulation occurred by postoperative day 1 and the patient was discharged by postoperative day 3. On the second day after the operation, CT and 3D reconstruction of the lower lumbar spine were performed for reexamination, which indicated that the severed bone had been completely removed (Figure 3). The postoperative effect was satisfactory, and the patient returned to normal operation one week after the operation.
Discussion
Posterior edge separation of the vertebral body is an uncommon but non-rare orthopedic disease (2-9), the cause of which is controversial. Previous studies (9,10) indicate that congenital dysplasia of vertebral epiphyseal may be the basis for the occurrence of this disease. Based on this, chronic and acute stresses such as excess weight, prolonged standing, and intense physical activity can act on the posterior edge of the vertebral body and cause it to move back, eventually pressing the spinal cord/nerve root to produce corresponding clinical symptoms. Posterior edge separation of the vertebral body is divided into 3 types (11): type I, cortical detachment at the posterior margin of the vertebral body; type II, avulsion fractures of the posterior margin of the vertebral body, including cortical and cancellous bones; type III, a bone mass near one side is detached, accompanied with a bone defect (the volume of the bone mass is smaller than that of the vertebral defect). In 1989, Epstein et al. (12) extended the parting to IV types: detachment of vertebral rear end plate alienation (involving the edge of the vertebral body length).
Similar to general spine surgical diseases, the treatment of posterior edge separation of the vertebral body can be simply classified into two categories. First, non-surgical treatment includes bed rest, non-steroidal anti-inflammatory drugs and physical therapy et al. (13). When non-surgical treatment fails, the second type of lumbar surgical treatment could be considered. Current lumbar surgeries involve posterior laminectomy decompression (14), fusion internal fixation (15) and minimally invasive surgery (16).
With the increasing popularization of minimally invasive technology, minimally invasive surgery with less trauma, less bleeding and faster recovery speed has been gradually recognized and promoted by patients and orthopedic surgeons (17-21). From September 2015 to May 2019, a total of 18 patients with Posterior edge separation of the vertebral body were treated in our department, all of whom were treated with minimally invasive techniques (9 cases of PELD/9 cases of MISS-TLIF), and the postoperative effects were satisfactory. From the previous experience in the treatment of lumbar disc herniation, we believe that the two minimally invasive techniques of intervertebral foramina and transforaminal intervertebral fusion are both suitable for the treatment of posterior edge separation of the vertebral body. Intervertebral foramoscope technology reaches the surgical area through a 7 mm surgical channel. With the auxiliary support of the external lens, it not only ensures a clear surgical field of view, but also bypasses the tissue interference such as dura sac and nerve root through the intervertebral foramina, where trephine and endoscopic bone knife are used to remove the broken bone. When faced with the imaging examination results indicating that the detached bone mass is large and strongly adherent to the dural sac, patients with small joint hyperplasia and cohesion, lateral recess stenosis, lumbar body instability or other degenerative changes, the minimally invasive transforaminal interbody fusion technique can be considered as an alternative treatment, in which the free bone mass is fully removed and the compression of the spinal canal is relieved, and some problems such as proliferating facet joints/lumbar instability can be handled simultaneously. Due to local anesthesia and less damage to the normal physiological structure of patients during operation, such as undisturbed inferior articular process, unresected cartilage final version and insertion of a pedicle screw; the PELD group has Less intraoperative blood loss and the shorter bed time after surgery.
Thus it can not only ensure the effect of operation at the time, but also avoid the influence of these degenerative diseases on the long-term satisfaction of the operation.
Among the many degenerative diseases of the spine, the incidence and diagnosis rate of the posterior edge separation of the vertebral body is low, so there are not many comprehensive clinical reports on the minimally invasive diagnosis and treatment of such diseases. It is believed that with the continuously in-depth research on the posterior edge separation of the vertebral body, and with the gradual deepening and popularization of the minimally invasive technique of the spine from the concept to the practice, the minimally invasive technique of the spine will bring light to patients with posterior edge separation of the vertebral body, better removing the disease and relieving the pain for the majority of patients!
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/apm-20-928
Data Sharing Statement: Available at http://dx.doi.org/10.21037/apm-20-928
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/apm-20-928). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Written informed consent for the experimental studies was obtained from the patients and all experiments were approved by the Institutional Ethics Committee of Third Affiliated Hospital of Jinzhou, Medical University.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Skobowytsh-Okolot B. Posterior Apophysis in L. IV--The Cause of Neuroradicular Disturbance. Acta Orthop Scand 1962;32:341-51. [Crossref] [PubMed]
- Akhaddar A, Boucetta M. Combined lumbar retroisthmic cleft and posterior ring apophysis separation in an adult. Spine J 2012;12:716-7. [Crossref] [PubMed]
- Xu DS, Walker CT, Godzik J, et al. Minimally invasive anterior, lateral, and oblique lumbar interbody fusion: a literature review. Ann Transl Med 2018;6:104. [Crossref] [PubMed]
- Lowrey JJ. Dislocated lumbar vertebral epiphysis in adolescent children. Report of three cases. J Neurosurg 1973;38:232-4. [Crossref] [PubMed]
- Kosztowski TA, Choi D, Fridley J, et al. Lumbar disc reherniation after transforaminal lumbar endoscopic discectomy. Ann Transl Med 2018;6:106. [Crossref] [PubMed]
- Deleo T, Merotto S, Smith C, et al. A posterior ring apophyseal fracture and disc herniation in a 21-year-old competitive basketball player: a case report. J Can Chiropr Assoc 2015;59:373-82. [PubMed]
- Wu XY, Ma W. Posterior lumbar ring apophysis fracture. Orthop Surg 2011;3:72-7. [Crossref] [PubMed]
- Pimenta L, Tohmeh A, Jones D, et al. Rational decision making in a wide scenario of different minimally invasive lumbar interbody fusion approaches and devices. J Spine Surg 2018;4:142-55. [Crossref] [PubMed]
- Akhaddar A, Belfquih H, Oukabli M, et al. Posterior ring apophysis separation combined with lumbar disc herniation in adults: a 10-year experience in the surgical management of 87 cases. J Neurosurg Spine 2011;14:475-83. [Crossref] [PubMed]
- Epstein NE, Epstein JA. Limbus lumbar vertebral fractures in 27 adolescents and adults. Spine (Phila Pa 1976) 1991;16:962-6. [Crossref] [PubMed]
- Takata K, Inoue S, Takahashi K, et al. Fracture of the posterior margin of a lumbar vertebral body. J Bone Joint Surg Am 1988;70:589-94. [Crossref] [PubMed]
- Epstein NE, Epstein JA, Mauri T. Treatment of fractures of the vertebral limbus and spinal stenosis in five adolescents and five adults. Neurosurgery 1989;24:595-604. [Crossref] [PubMed]
- Gautschi OP, Hildebrandt G, Cadosch D. Acute low back pain--assessment and management. Praxis (Bern 1994) 2008;97:58-68.
- Overdevest G, Vleggeert-Lankamp C, Jacobs W, et al. Effectiveness of posterior decompression techniques compared with conventional laminectomy for lumbar stenosis. Eur Spine J 2015;24:2244-63. [Crossref] [PubMed]
- Sun Y, Zhang W, Qie S, et al. Comprehensive comparing percutaneous endoscopic lumbar discectomy with posterior lumbar internal fixation for treatment of adjacent segment lumbar disc prolapse with stable retrolisthesis: A retrospective case-control study. Medicine (Baltimore) 2017;96:e7471. [Crossref] [PubMed]
- Koebbe CJ, Maroon JC, Abla A, et al. Lumbar microdiscectomy: a historical perspective and current technical considerations. Neurosurg Focus 2002;13:E3. [Crossref] [PubMed]
- Staub BN, Sadrameli SS. The use of robotics in minimally invasive spine surgery. J Spine Surg 2019;5:S31-S40. [Crossref] [PubMed]
- Sharif S, Afsar A. Learning Curve and Minimally Invasive Spine Surgery. World Neurosurg 2018;119:472-8. [Crossref] [PubMed]
- Yoo JS, Parrish JM, Jenkins NW, et al. Postoperative Outcomes Based on American Society of Anesthesiologists Score After Minimally Invasive Transforaminal Lumbar Interbody Fusion. Clin Spine Surg 2020;33:E40-2. [Crossref] [PubMed]
- Byvaltsev VA, Kalinin AA, Konovalov NA. Minimally invasive spinal surgery: stages of development. Zh Vopr Neirokhir Im N N Burdenko 2019;83:92-100. [Crossref] [PubMed]
- Li H, Jiang C, Mu X, et al. Comparison of MED and PELD in the Treatment of Adolescent Lumbar Disc Herniation: A 5-Year Retrospective Follow-Up. World Neurosurg 2018;112:e255-60. [Crossref] [PubMed]


