
Drug treatment of ankylosing spondylitis and related complications: an overlook review
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory disease characterized by back pain and progressive spinal stiffness. The patient may have multiple joint involvement, followed by spinal, sacroiliac joint and hip pain and joint deformity. As a result, the patient’s quality of life will be seriously affected (1,2). What is more, AS is a common disease in young adults, with a peak age of onset in their 20s to 30s. A recent survey found that the prevalence of AS is 30 per 10,000 people. With the change of living environment and life-style, there is an increase of incidence of AS (3). However, the pathogenesis of AS is still controversial. Genetic factors may be involved in the pathogenesis of the disease. A survey found that the prevalence rate of AS in HLA-B27 positive people is about 5–6% (4). In fact, human leukocyte antigen-B27 (HLA-B27), gut microbiome, innate-like lymphoid cells and mechanical stress are important contributors. Tumor necrosis factor-alpha (TNF-α) and interleukin-17A (IL-17A) are major mediators involved in pathology (2,5). In addition, inflammation and autoimmune associated inflammation may lead to the occurrence and development of AS (5,6). Therefore, biological agents for inflammatory factors is not only the frontier of AS therapy, but also the direction of new drug research and development (7).
At present, the first line choice for pain relieve is nonsteroidal anti-inflammatory drugs (NSAIDs). Patients with milder symptoms can choose disease-modifying antirheumatic drugs (DMARDs) or biological agents. However, the efficacy of these drugs in relieving AS is limited. In addition, there is heterogeneity in drug responses in different patients. Furthermore, AS patients often have complications or co-existing diseases, which may affect the choice of drugs and the treatment effect (8,9). Firstly, patients with AS may be complicated with uveitis, inflammatory bowel disease (IBD), or psoriasis, which usually have typical symptoms (10). What is more, AS patients are prone to cardiovascular diseases, which are promoted by the occurrence and development of atherosclerosis, a disease would also increase the risk of stroke (11,12). A latest multivariate analysis of patients with AS found that these people had a higher risk of developing pulmonary embolism (PE) and deep venous thrombosis (DVT). In addition, the hazard ratio (HR) in the first year was 2.8 and 2.2 respectively. Therefore, early diagnosis of AS and early intervention may reduce the risk of venous thromboembolism (VTE) in patients with AS (13). At the same time, there is evidence that AS patients are prone to diabetes mellitus (DM) and promote the occurrence and development of osteoporosis (14,15). Furthermore, AS patients may have intestinal flora abnormality, resulting in abnormal physiological state of the body and a variety of diseases (16). In fact, these complications and co-existing diseases are likely to be related to a poor prognosis of AS patients and even promote AS (15). Therefore, the reasonable selection of drugs and multidisciplinary diagnosis and treatment of co-existing diseases are of great significance, which may improve the therapeutic effect and long-term prognosis of AS (17,18).
An increasing attention has been paid to the complications and the co-exist chronic diseases of AS. However, besides patient’s condition and physician experience, the cost, benefits and potential side effects of the drugs need to be taken into account during the selection of drug therapies. Therefore, the reasonable choice of drugs is still worth exploring. Through retrospective investigation and analysis, Li et al. found that multidisciplinary diagnosis and treatment of AS can reduce the delay in the diagnosis of AS and improve the prognosis (18). This paper briefly summarizes the recent clinical drug treatment, the possible complications of AS, as well as the selection of multidisciplinary drug therapy and other matters needing attention. We hope to provide ideas and clues for the rational use of drugs and synergistic treatment of AS in order to improve the clinical therapeutic effect.
Drugs commonly used in the treatment of AS
The goal of AS treatment is to improve the effect of treatment, and improve the quality of life of patients. Therefore, it is necessary to alleviate the symptoms such as pain and limitation of activity as far as possible, reduce the occurrence of extraarticular lesions or coexisting diseases, and maintain the psychosocial function of the patients to the maximum extent and delay the progress of the disease (19). In fact, drug therapy is still the main means of AS treatment and is effective in improving symptoms. At the same time, it may accompany with gastrointestinal and cardiovascular side effects (2).
Nonsteroidal anti-inflammatory drugs (NSAIDs)
NSAIDs are still the first choice for AS treatment and can effectively relieve pain. There is evidence that many patients only need NSAIDs to achieve the treatment goal. It is estimated that 70–80% of AS patients have significant relief of back pain and stiffness after NSAIDs treatment (20,21). Commonly used drugs include ibuprofen, celecoxib and naproxen. However, the gastrointestinal, cardiovascular and other side effects caused by the use of these drugs need to be considered (22). In addition, attention should be paid to the regular use of these drugs. Furthermore, whether the use of NSAID in asymptomatic AS patients is effective in improving the condition remains to be proved by further clinical trials (23,24).
Disease-modifying antirheumatic drugs (DMARDs)
Conventional synthetic DMARDs can alleviate and improve the symptoms to some extent, however, it usually takes months. Commonly used drugs include sulfasalazine (SSZ), methotrexate (MTX) and thalidomide (25,26). SSZ can inhibit leukocyte migration, reduce proteolytic enzyme activity, and inhibit the effect of a variety of cytokines, which is effective in early diagnosed of young patients (27). However, a new guideline suggests that SSZ is recommended only for patients with persistent peripheral arthritis prohibited by TNF inhibitors (2). MTX is a folic acid antagonist, which inhibits the expression of various cytokines by blocking the synthesis of DNA (28). Thalidomide improves clinical symptoms by regulating immunity and inhibiting TNF- α gene expression. However, there is evidence that it may cause peripheral neuropathy (29). Despite the limited therapeutic effect and side effects of DMARDs, the low cost and the ability to improve certain symptoms are its advantages. In addition, biological DMARDs are highly effective drugs in achieving remission (30). The benefits of the combined use of these drugs need to be further clarified (2).
Targeted biological agents
This kind of drugs usually take effect quickly and have good effects. At present, it is widely used in practice, and also a promised direction of new drug research and development (2). The most commonly used drugs are TNF-α inhibitors, including infliximab and cetuzumab. In fact, the use of TNF-α inhibitors can often improve the symptoms when the effect of NSAIDs are far from satisfactory. What is more, TNF inhibitors are also effective in patients with advanced AS (31). Another commonly used biological agent category is the IL-17 monoclonal antibody drugs. For example, secukinumab is an anti-IL-17 monoclonal antibody, which has the similar curative effect with TNF inhibitors. However, secukinumab is the preferred biological agent for high risk patients with tuberculosis (TB) since TNF inhibitors may reactivate latent TB (32). In addition, secukinumab and other anti-IL-17 biological agents may induce or aggravate IBD (33). These are the problems that should be paid more attention to in clinical drug selection. Ixekizumab is another monoclonal antibody against IL-17. It is commonly used in the treatment of psoriasis and psoriatic arthritis, and its clinical evidence for the improvement of AS remains to be clarified (34). Furthermore, the efficacy and safety of Janus kinase (JAK) 1 inhibitor and other biological agents need to be observed in further clinical trials (35).
Common complications or coexisting diseases
Cardiovascular diseases
There is evidence that patients with AS are more likely to have cardiovascular diseases. AS may lead to the occurrence and development of cardiovascular diseases and increase the risk of stroke. At the same time, it is also associated with aortic valve insufficiency and acute coronary syndrome (12). A cohort study found that patients with AS are more likely to develop venous thrombosis, with a HR of 1.53 (13). A meta-analysis of AS and subclinical atherosclerosis suggested that AS may promote the occurrence and development of atherosclerosis (11). In addition, a group survey found that there was a correlation between AS and atrial fibrillation. AS was an independent risk factor for atrial fibrillation, suggesting that patients with AS need to prevent complications such as thrombosis at an early stage (36). Furthermore, the use of AS drugs such as NSAIDs is also a cause of cardiovascular disease (12). Therefore, early cardiovascular disease-related monitoring and prevention in patients with AS can minimize complications.
Arthritis, osteoporosis and fracture
AS patients are prone to arthritis, which may involve several joints, as a result, seriously reducing the quality of life. NSAIDs may relieve pain, but it has limited effect on arthritis. On the contrary, DMARDs such as SSZ have good effects on the improvement of peripheral joint disease (2). AS patients are also prone to osteoporosis, which can easily lead to bone fracture. What is more, this situation is obvious in the first 10 years of the course of the disease (15,37). Furthermore, AS patients are prone to fracture, resulting nerve injury, pneumonia, thrombus and other fracture-related complications (38). A survey found that patients with AS are much more likely to develop spinal cord injury than the general population, and that the cervical spine is the most frequently affected (39). Since 65% of spinal fractures are associated with neurological complications, the patient’s initial complaint is usually mild and may be ignored. It is worth noting that even minor injuries can lead to vertebral fracture and spinal cord injury in patients with AS (40). Therefore, patients with AS should pay more attention to the early treatment of arthritis and osteoporosis.
Staphylococcal inflammation, IBD and psoriasis
Due to the obvious symptoms of some patients, there are many related reports. The prevalence of staphylococcitis in patients with AS was higher than that in the general population. A meta-analysis suggested that unilateral anterior uveitis was the most common extraarticular complication of axial spondyloarthritis, with an incidence of 25–40% (41). A study found that TNF inhibitor therapy may effectively reduce the frequency and recurrence rate of uveitis in patients with AS (42). In addition, AS patients are prone to have an increased incidence of IBD. It is estimated that about 5–10% of AS patients have dominant IBD, and the presence of IBD may affect the choice of therapeutic drugs (41,43). Furthermore, according to the investigation, about 10% AS patients complicated with psoriasis (41). In fact, AS patients with psoriasis are more likely to have peripheral joint involvement than those without psoriasis. In addition, AS patients with psoriasis tend to have more severe symptoms (44). Therefore, the reasonable selection and use of drugs may affect the prognosis of patients.
Others
AS may cause pain, fatigue, decreased sleep quality, which affecting mental health, inducing depression, and reducing quality of life. A survey of 314 patients with AS found that 184 (58.6%) had a high incidence of sleep disorders (45,46). In fact, psychosomatic factors may affect the overall quality and long-term prognosis of AS. Therefore, in addition to controlling disease activity, attention should also be paid to anxiety, depression, sleep disorders and sense of helplessness in patients with AS (47). Furthermore, there is a correlation and interrelationship between AS and intestinal flora abnormality (16). More and more attention has been paid to the importance of intestinal flora in maintaining body health. Intestinal flora may be involved in the occurrence and development of depression, IBD, osteoporosis and other diseases (48,49). Therefore, the relationship between AS and intestinal flora is worthy of further discussion.
Multidisciplinary combined therapy
Advantages
Heterogeneity exists in patients with AS. In fact, different patients have various conditions, reactivity to drugs, complications or coexisting diseases. Multidisciplinary therapy (MDT) is of great value in drug selection, reasonable combination, and the greatest benefit in improving the prognosis of AS. TNF inhibitors are currently the most effective treatment for AS, but may be resistant or ineffective to some of the patients. A number of patients who were treated with TNF inhibitors for weeks to months has no improvement of the symptoms. Some patients experienced an initial remission, however worsened later (19,50). Besides further search for specific therapeutic targets for AS (51), multidisciplinary diagnosis and treatment may be most beneficial to a better management. Most treatments with different clinical manifestations overlap, while some therapeutic drugs are more effective for certain characteristics. Furthermore, health care providers in different disciplines have their own best judgment and drug treatment experience. Therefore, multidisciplinary consultation and cooperation can help to choose the most appropriate strategy (52) (Figure 1).
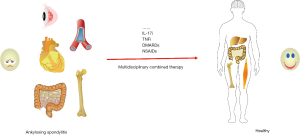
Attentions
Patients with AS need an appropriate time point for the initiation of combined medication. For patients with mild early symptoms, the use of NSAIDs is effective. At the same time, the sequence of medication is also important. Patients with peripheral arthritis are usually treated with SSZ or methotrexate. The early use of biological agents needs to consider the performance-to-price ratio. Furthermore, the development of AS is a dynamic process, we need to monitor the progress of the disease and choose the most appropriate drugs for complications (19).
Summary and prospect
The treatment of AS and its related complications is still a filed worth exploring. In fact, the heterogeneity of AS is significant for the selection of therapeutic drugs. Heterogeneity is mainly due to differences in pathology and etiology. MDT for AS has important value and development prospect. The choice of specific drug combinations requires further clinical evidence. Targeted drugs for inflammatory factors are the frontier of treatment, but the cost and safety of treatment still need to be considered. Furthermore, the research and development of multi-target therapeutic drugs has a broad prospect as a variety of mechanisms may be involved in AS and its co-existing diseases. In general, the poor prognosis and complications of patients are often related to the duration of the disease. Therefore, it is necessary to further study the molecular targets for early diagnosis of AS.
Acknowledgments
We are thankful to Xi Chen for her assistance with the suggestions during the submission process.
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/apm-20-277). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Rudwaleit M, van der Heijde D, Khan MA, et al. How to diagnose axial spondyloarthritis early. Ann Rheum Dis 2004;63:535-43. [Crossref] [PubMed]
- Ward MM, Deodhar A, Gensler LS, et al. 2019 Update of the American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network Recommendations for the Treatment of Ankylosing Spondylitis and Nonradiographic Axial Spondyloarthritis. Arthritis Rheumatol 2019;71:1599-613. [Crossref] [PubMed]
- Wang R, Ward MM. Epidemiology of axial spondyloarthritis: an update. Curr Opin Rheumatol 2018;30:137-43. [Crossref] [PubMed]
- Dean LE, Jones GT, MacDonald AG, et al. Global prevalence of ankylosing spondylitis. Rheumatology (Oxford) 2014;53:650-7. [Crossref] [PubMed]
- Gravallese EM, Schett G. Effects of the IL-23-IL-17 pathway on bone in spondyloarthritis. Nat Rev Rheumatol 2018;14:631-40. [Crossref] [PubMed]
- Vanaki N, Aslani S, Jamshidi A, et al. Role of innate immune system in the pathogenesis of ankylosing spondylitis. Biomed Pharmacother 2018;105:130-43. [Crossref] [PubMed]
- Qaiyum Z, Gracey E, Yao Y, et al. Integrin and transcriptomic profiles identify a distinctive synovial CD8+ T cell subpopulation in spondyloarthritis. Ann Rheum Dis 2019;78:1566-75. [Crossref] [PubMed]
- Albrecht K, Callhoff J, Zink A. Long-term trends in rheumatology care: Achievements and deficits in 25 years of the German national rheumatology database. Z Rheumatol 2019;78:703-12. [Crossref] [PubMed]
- Smolen JS. Treat to Target in Rheumatology: A Historical Account on Occasion of the 10th Anniversary. Rheum Dis Clin North Am 2019;45:477-85. [Crossref] [PubMed]
- Varkas G, Vastesaeger N, Cypers H, et al. Association of Inflammatory Bowel Disease and Acute Anterior Uveitis, but Not Psoriasis, With Disease Duration in Patients With Axial Spondyloarthritis: Results From Two Belgian Nationwide Axial Spondyloarthritis Cohorts. Arthritis Rheumatol 2018;70:1588-96. [Crossref] [PubMed]
- Bai R, Zhang Y, Liu W, et al. The Relationship of Ankylosing Spondylitis and Subclinical Atherosclerosis: A Systemic Review and Meta-Analysis. Angiology 2019;70:492-500. [Crossref] [PubMed]
- Eriksson JK, Jacobsson L, Bengtsson K, et al. Is ankylosing spondylitis a risk factor for cardiovascular disease, and how do these risks compare with those in rheumatoid arthritis. Ann Rheum Dis 2017;76:364-70. [Crossref] [PubMed]
- Avina-Zubieta JA. Risk of venous thromboembolism in ankylosing spondylitis: a general population-based study. Ann Rheum Dis 2019;78:480-5. [Crossref] [PubMed]
- Liao KF, Kuo YH, Lai SW, et al. Diabetes mellitus in ankylosing spondylitis. Ann Rheum Dis 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Walsh JA, Song X, Kim G, et al. Evaluation of the comorbidity burden in patients with ankylosing spondylitis using a large US administrative claims data set. Clin Rheumatol 2018;37:1869-78. [Crossref] [PubMed]
- Zhang L, Hu Y, Xu Y, et al. The correlation between intestinal dysbiosis and the development of ankylosing spondylitis. Microb Pathog 2019;132:188-92. [Crossref] [PubMed]
- Liang L, Pan Y, Wu D, et al. Effects of Multidisciplinary Team-Based Nurse-led Transitional Care on Clinical Outcomes and Quality of Life in Patients With Ankylosing Spondylitis. Asian Nurs Res (Korean Soc Nurs Sci) 2019;13:107-14. [Crossref] [PubMed]
- Li J, Xu Y, Chen Y, et al. A multidisciplinary clinic approach to improve physician-related diagnostic delay for patients with axial spondyloarthritis: a retrospective study. J Int Med Res 2019;47:2483-91. [Crossref] [PubMed]
- van der Heijde D, Ramiro S, Landewé R, et al. 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann Rheum Dis 2017;76:978-91. [Crossref] [PubMed]
- Song IH, Poddubnyy DA, Rudwaleit M, et al. Benefits and risks of ankylosing spondylitis treatment with nonsteroidal antiinflammatory drugs. Arthritis Rheum 2008;58:929-38. [Crossref] [PubMed]
- Varkas G, Jans L, Cypers H, et al. Brief Report: Six-Week Treatment of Axial Spondyloarthritis Patients With an Optimal Dose of Nonsteroidal Antiinflammatory Drugs: Early Response to Treatment in Signal Intensity on Magnetic Resonance Imaging of the Sacroiliac Joints. Arthritis Rheumatol 2016;68:672-8. [Crossref] [PubMed]
- Kroon FP, van der Burg LR, Ramiro S, et al. Nonsteroidal Antiinflammatory Drugs for Axial Spondyloarthritis: A Cochrane Review. J Rheumatol 2016;43:607-17. [Crossref] [PubMed]
- Kroon F, Landewe R, Dougados M, et al. Continuous NSAID use reverts the effects of inflammation on radiographic progression in patients with ankylosing spondylitis. Ann Rheum Dis 2012;71:1623-9. [Crossref] [PubMed]
- Sieper J, Listing J, Poddubnyy D, et al. Effect of continuous versus on-demand treatment of ankylosing spondylitis with diclofenac over 2 years on radiographic progression of the spine: results from a randomised multicentre trial (ENRADAS). Ann Rheum Dis 2016;75:1438-43. [Crossref] [PubMed]
- Chan J, Gladman D. Oral treatment options for AS and PsA: DMARDs and small-molecule inhibitors. Best Pract Res Clin Rheumatol 2018;32:415-26. [Crossref] [PubMed]
- D'Angelo S, Carriero A, Gilio M, et al. Safety of treatment options for spondyloarthritis: a narrative review. Expert Opin Drug Saf 2018;17:475-86. [Crossref] [PubMed]
- Khanna Sharma S, Kadiyala V, Naidu G, et al. A randomized controlled trial to study the efficacy of sulfasalazine for axial disease in ankylosing spondylitis. Int J Rheum Dis 2018;21:308-14. [Crossref] [PubMed]
- Cipriani P, Ruscitti P, Carubbi F, et al. Methotrexate: an old new drug in autoimmune disease. Expert Rev Clin Immunol 2014;10:1519-30. [Crossref] [PubMed]
- Xue HX, Fu WY, Cui HD, et al. High-dose thalidomide increases the risk of peripheral neuropathy in the treatment of ankylosing spondylitis. Neural Regen Res 2015;10:814-8. [Crossref] [PubMed]
- Cantini F, Niccoli L, Nannini C, et al. Second-line biologic therapy optimization in rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. Semin Arthritis Rheum 2017;47:183-92. [Crossref] [PubMed]
- Dougados M, Braun J, Szanto S, et al. Efficacy of etanercept on rheumatic signs and pulmonary function tests in advanced ankylosing spondylitis: results of a randomised double-blind placebo-controlled study (SPINE). Ann Rheum Dis 2011;70:799-804. [Crossref] [PubMed]
- Kammuller M, Tsai TF, Griffiths CE, et al. Inhibition of IL-17A by secukinumab shows no evidence of increased Mycobacterium tuberculosis infections. Clin Transl Immunology 2017;6:e152. [Crossref] [PubMed]
- Hohenberger M, Cardwell LA, Oussedik E, et al. Interleukin-17 inhibition: role in psoriasis and inflammatory bowel disease. J Dermatolog Treat 2018;29:13-8. [Crossref] [PubMed]
- Kiwalkar S, Beier S, Deodhar A. Ixekizumab for treating ankylosing spondylitis. Immunotherapy 2019;11:1273-82. [Crossref] [PubMed]
- van der Heijde D, Baraliakos X, Gensler LS, et al. Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active ankylosing spondylitis (TORTUGA): results from a randomised, placebo-controlled, phase 2 trial. Lancet 2018;392:2378-87. [Crossref] [PubMed]
- Moon I, Choi EK, Jung JH, et al. Ankylosing spondylitis: A novel risk factor for atrial fibrillation - A nationwide population-based study. Int J Cardiol 2019;275:77-82. [Crossref] [PubMed]
- Geusens P, Lems WF. Osteoimmunology and osteoporosis. Arthritis Res Ther 2011;13:242. [Crossref] [PubMed]
- Miao J, Chen Y, Zhang B, et al. Surgical Treatment for Odontoid Fractures in Patients with Long-Standing Ankylosing Spondylitis: A Report of 3 Cases and Review of the Literature. World Neurosurg 2018;116:88-93. [Crossref] [PubMed]
- Chaudhary SB, Hullinger H, Vives MJ. Management of acute spinal fractures in ankylosing spondylitis. ISRN Rheumatol 2011;2011:150484. [Crossref] [PubMed]
- Thumbikat P, Hariharan RP, Ravichandran G, et al. Spinal cord injury in patients with ankylosing spondylitis: a 10-year review. Spine (Phila Pa 1976) 2007;32:2989-95. [Crossref] [PubMed]
- Stolwijk C, van Tubergen A, Castillo-Ortiz JD, et al. Prevalence of extra-articular manifestations in patients with ankylosing spondylitis: a systematic review and meta-analysis. Ann Rheum Dis 2015;74:65-73. [Crossref] [PubMed]
- van Denderen JC, Visman IM, Nurmohamed MT, et al. Adalimumab significantly reduces the recurrence rate of anterior uveitis in patients with ankylosing spondylitis. J Rheumatol 2014;41:1843-8. [Crossref] [PubMed]
- El MA. Extra-articular manifestations of ankylosing spondylitis: prevalence, characteristics and therapeutic implications. Eur J Intern Med 2011;22:554-60. [Crossref] [PubMed]
- Perez AR, Maldonado CJA, Citera G, et al. Differential features between primary ankylosing spondylitis and spondylitis associated with psoriasis and inflammatory bowel disease. J Rheumatol 2011;38:1656-60. [Crossref] [PubMed]
- Li Y, Zhang S, Zhu J, et al. Sleep disturbances are associated with increased pain, disease activity, depression, and anxiety in ankylosing spondylitis: a case-control study. Arthritis Res Ther 2012;14:R215. [Crossref] [PubMed]
- Brionez TF, Assassi S, Reveille JD, et al. Psychological correlates of self-reported disease activity in ankylosing spondylitis. J Rheumatol 2010;37:829-34. [Crossref] [PubMed]
- Jang JH, Green CE, Assassi S, et al. The contribution of disease activity on functional limitations over time through psychological mediators: a 12-month longitudinal study in patients with ankylosing spondylitis. Rheumatology (Oxford) 2011;50:2087-92. [Crossref] [PubMed]
- Kc D, Sumner R, Lippmann S. Gut microbiota and health. Postgrad Med 2020;132:274. [Crossref] [PubMed]
- Hao ML, Wang GY, Zuo XQ, et al. Gut microbiota: an overlooked factor that plays a significant role in osteoporosis. J Int Med Res 2019;47:4095-103. [Crossref] [PubMed]
- Hamilton L, Barkham N, Bhalla A, et al. BSR and BHPR guideline for the treatment of axial spondyloarthritis (including ankylosing spondylitis) with biologics. Rheumatology (Oxford) 2017;56:313-6. [Crossref] [PubMed]
- Quaden DH, De Winter LM, Somers V. Detection of novel diagnostic antibodies in ankylosing spondylitis: An overview. Autoimmun Rev 2016;15:820-32. [Crossref] [PubMed]
- van Mens LJJ, van de Sande MGH. DLP B. New treatment paradigms in spondyloarthritis. Curr Opin Rheumatol 2018;30:79-86. [Crossref] [PubMed]


