
Nutrition counseling combined with head and neck rehabilitation exercises can enhance outcomes among nasopharyngeal carcinoma patients in southern China: a prospective study in an epidemic area
Introduction
Chronic diseases like cancer impose an enormous and growing burden on individuals, families, and society, as well as on the health care systems in the United States and throughout the world (1). Nasopharyngeal carcinoma (NPC) is a cancer arising from the nasopharyngeal epithelium with 71% of new cases occurring in the east and southeast parts of Asia, with South-Central Asia and North and East Africa accounting for the remainder. However, NPC is rare in the Western world (2). As a consequence, the development of intervention programs that can educate and assist people in adopting and maintaining long-term behavioral changes to prevent further progression of the disease and improve quality of life represents a continuing need (1,3,4).
A survey from Germany revealed a huge gap between the need and actual delivery of nutritional care services (4-7) (https://www.selfmanagementresource.com/). The program has been replicated in diverse populations, including in China, and has been found to improve healthy behavior, self-efficacy, health status, and number of hospitalizations (8-10). Recent studies have shown that self-management interventions can improve some of the physical and psychosocial problems associated with cancer survivorship and suggested that self-management programs for cancer survivors may be particularly beneficial during the transition period from primary treatment to longer-term survivorship (11,12). After primary treatment, many cancer survivors are left to deal with physical and psychosocial problems such as fatigue, pain, functional limitations, anxiety, depression, and decreased quality of life, and may have no way to find help (13).
In the current study, we focused on NPC patient nutrition counseling and rehabilitation training delivered by nurses, and the effect of such counseling on responses to the CDSM questionnaire (14). We compared the mean change in the CDSM index between an intervention group and a control group. We also compared the change in the CDSM index mean between the two groups before treatment, immediately after treatment, and 3, 6, and 12 months after treatment. The aim of the current study was to study the effect of nutritional care and rehabilitation training of the head and neck on CDSM indicators (including self-reported global health, the daily effects of illness, fatigue, shortness of breath, pain, stretching/strengthening exercises, aerobic exercises, cognitive symptom management, communication with doctors, and self-efficacy) in NPC patients receiving IMRT.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/apm-20-1053).
Methods
Patients
Patients diagnosed with undifferentiated NPC, with primary tumor stages I–IV as per the International Union Against Cancer (UICC) tumor-node-metastasis (TNM) tumor classification, were prospectively enrolled for treatment with curative intention. The patients received IMRT at Sun Yat-sen University Cancer Center (SYSUCC) between November 2013 and February 2015. The eligibility criteria for the patients included the following: (I) diagnosis of primary untreated NPC; (II) age between 18 and 65 years; (III) agreement to participate in the study and provide written informed consent; and (IV) completion of the questionnaires by the patients themselves. The completed questionnaires were returned to the research nurses for subsequent data entry. Patients with a history of head and neck irradiation or surgery were excluded from the study. In addition, the study also excluded patients who had severe disease of the liver, heart, lung, or kidney; dysnomia; or impaired mental status. The criteria has been explained previously (14,15).
The study protocol was approved by the Research Ethics Committee of SYSUCC, Guangzhou, China (No. B201502401). Demographic data concerning gender, age, educational status, marital status, treatment, and UICC stage were obtained. The CDSM questionnaire was administered at 5 time points: pretreatment, immediately after treatment, and 3, 6, and 12 months after IMRT.
Treatment
All patients (141 patients) were treated using IMRT. All target volumes were depicted slice-by-slice on the treatment planning computed tomography (CT) scan. The primary nasopharyngeal gross tumor volume (GTV nx) and the involved cervical lymph nodes were determined based on imaging, clinical, and endoscopic findings. The enlarged retropharyngeal nodes together with the primary GTV were outlined as the GTV nx on the IMRT plans. The first clinical tumor volume (CTV1) was defined as the area within 0.5–1.0 cm outside the GTV, which is a site that involves potential sites of local infiltration. The clinical target volume 2 (CTV2) was defined as the margin from 0.5–1.0 cm around CTV1 and the lymph node draining area (levels II, III, and IV). The IMRT doses in the target region were 69.96–73.92 Gy for the GTV nx, 69.96–73.92 Gy for the GTV nd, 60–66 Gy for the PTV-1, and 54–56 Gy for the PTV-2, with treatment plans being varied in accordance with the TV and cancer stage. The IMRT dose was 2.0–2.2 Gy per fraction per day. Irradiation was performed 5 times a week, 1 time a day, and 2 days of rest were taken. The total number of exposures was 30–33. The total dose of IMRT for NPC patients followed the guidelines of SYSUCC (16). Chemotherapy in the current study included induction and concomitant chemotherapies as explained previously (17). The induction chemotherapy regimen comprised paclitaxel (135–175 mg/m2), fluorouracil (500–750 mg/m2), and cisplatin (60–90 mg/m2) (TPF) every 3 weeks for 2–3 cycles, while concurrent chemotherapy regimen included dipeptidyl-peptidase (DDP) (80–100 mg/m2) every 3 weeks for 2–3 cycles.
Questionnaire
The CDSM was measured at 5 time points: pretreatment, immediately after treatment, and 3, 6, and 12 months after IMRTee. It included the 44 following questions and respective calculations: (I) 10 items of self-rated global health, 3 of which were reversed, with the score being averaged; (II) 10 items of the daily effects of illness, with the score being averaged; (III) each of fatigue, shortness of breath, and pain as single items; (IV) stretching/strengthening exercise (minutes/week), calculated as exercise frequency multiplied by time; (V) aerobic exercise (minutes/week) comprising 5 items, calculated as the total minutes of the items; (VI) cognitive symptom management comprising 6 items, with the score being averaged; (VII) communication with the physician comprising 3 items, with the score being averaged; (VIII) self-efficacy comprising 6 items, with the score being averaged.
Intervention methods
Patients in the control group received leaflets concerning nutrition and health and were provided with regular assistance. Patients in the intervention group were given a rehabilitation training manual and a video CD. The nutritional intervention was performed by nurses (qualified third-level public dieticians and a specialist nurse in swallowing) who were intensely trained in the regimen. They were generally instructed to provide sufficient energy, protein, micronutrients and fluids to meet nutritional requirements, thereby maintaining or improving nutritional status. Average energy requirement (EER) was used to ensure energy intake, and the recommended energy requirement for patients was 27–30 kcal/kg per day. The specifics of the nutritional intervention consisted of the following:
- Carbohydrates: accounting for 55–60% of total daily calories at 100–150 g/d;
- Fiber: at 30 g/d for males and 21 g/d for females;
- Fat: accounting for 25–30% of total calories per day; n-6 polyunsaturated fatty acids (linoleic acid) accounting for 5–10% of the total calories (14 g/d for male and 11 g/d for female); Omega-3 polyunsaturated fatty acids (a-linolenic acid) accounting for 0.6–1.2% of total daily calories (1.6 g/d for males and 1.1 g/d for females);
- Protein: accounting for 10–30% of total daily calories at 1.0–1.2 g/kg per day;
- Water: approximately 2.5 L/d for males and 2.0 L/d for female adjusted for specific conditions to be adjusted. Patients, family members and caregivers were considered nutrition education targets. The main contents of the intensive nutrition consultation education included four aspects: (i) food selection: appropriate and taboo food; (ii) food preparation: adjustment of diet texture; (iii) dietary guidance: dining environment, time and water consumption; (iv) use of leaflets, bedside guidance, reminder card for nutrition guidance, and help with providing meals according to need.
Evaluate the effectiveness of the education nutrition content, and the compliance of the patient and caregiver. Treatment of dental problems timely, restoration of teeth properly and installation of denture to make it easier to eat for patients. For patients with or at risk of malnutrition, oral supplements should be added on the basis of diet to improve the intake of energy, protein, and micronutrients, so as to maintain or improve the nutritional status. Patients with hyposmia, hypogeusia or anorexia, were given food of different flavors, textures, temperatures, and colors to stimulate appetite. Confirm the patients with dysphagia and dysphagia, and evaluate the swallowing function to change the food consistency and feeding mode. Patients with body mass index (BMI) <18.5 kg/m2 should be evaluated comprehensively and nutrition intervention should be implemented. For patients whose metabolic status or physiological functions are abnormal and cannot be treated with conventional methods, they should consult nutrition experts and develop nutrition support programs. Distribute dietary guidelines for patients with NPC after radiotherapy; outpatient follow-up and telephone follow-up.
Rehabilitation exercises of the head and neck
Relaxation massage: (I) massage of the masticatory muscle group: circular massage with both thumb on both cheeks near the molar from top to bottom; (II) jaw muscle group and Adam’s apple muscle group: with the head slightly back, use the thumb to massage gently, 5–15 minutes per time, 3–5 times.
Slow rhythmic movement of the temporomandibular joint: (I) 5 s of small, medium, and large mouth opening, each at an interval of 10 s, 10 times for 1 set, and a total of 5 sets; (II) fast rhythmic movement of the temporomandibular joint: 1 s of small, medium, and large mouth opening, each at an interval of 10 s, 10 times for 1 set, and a total of 5 sets.
Functional training for the neck and shoulder: (I) look at the abdomen by lowering the head: stand upright with eyes to the front, with feet and shoulders level, slowly look at the abdomen for 10 seconds at the maximum limit and slowly return to the original position; (II) look up and enjoy the moon: slowly look up to the maximum for 10 seconds and then slowly return to the original position; (III) look to the left and right: stand upright with eyes to the front, with feet and shoulders level; swivel the head to the left slowly, and rotate the neck to the maximum for 10 seconds; then, turn the head to the neutral position, then to the right for 10 seconds at the maximum limit, and slowly return to the original position; (IV) shake from left to right: stand upright with eyes to the front, with feet and shoulders level; slowly turn the head to the left until to the maximum for 10 seconds, and then turn the head to the neutral position; turn to the right side until the maximum and slowly return to the original position after a few seconds. Each action should be repeated 8 times, 3 times per day.
Shoulder exercises: place hands at shoulder height and turn the head to the right, with the palm of the right hand turning up and backward. The palm of the left hand turns down and backward keeping shoulders tight. Then switch sides, and repeat 10 times.
Outcomes
As of March 27, 2019, there were 21 patients who had distant metastasis. Of these patients, 2 had loco regional recurrence, 3 died, and 1 died with no distant metastasis or recurrence. In all, there were 22 ending events.
Statistical analysis
All analyses were performed using SPSS version 23.0 (version23.0; SPSS Inc., Chicago, II, USA). All tests were conducted with a two-sided 0.05 significance level. Descriptive statistics were used to analyze patient demographic data and clinical characteristics. Differences in the CDSM measurements at the 5 time points (pretreatment, post-treatment, and 3, 6, and 12 months after IMRT) were evaluated. An independent samples T test was used to compare the mean of the CDSM items and assess changes between the two groups. Non-parametric tests were performed when the data were not normally distributed.
Univariate analysis performed using the Cox proportional hazards regression model was used to calculate the P values associated with age, gender, nutritional support; levels of white blood cells, leukomonocytes, hemoglobin, thrombocytes, albumin, and c-reactive protein; and education, and T stage, N stage, American Joint Committee on Cancer (AJCC) stage; and each CDSM scale (self-rated global health, the daily effects of illness, fatigue, shortness of breath, pain, stretching/strengthening exercises, aerobic exercises, cognitive symptom management, communication with doctor, self-efficacy) (see Table 1). When the P value in the univariate analysis was less than 0.05, the scale was separately calculated through a multivariate analysis adjusted for age, gender, nutritional support; levels of white blood cells, leukomonocytes, hemoglobin, thrombocytes, albumin, and c-reactive protein; and education, T stage, N stage, AJCC stage, occupation, and marriage.
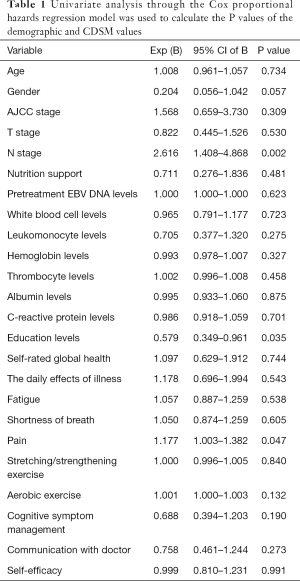
Full table
Results
Characteristics of the population
A total of 141 individuals were enrolled in the study (Table 2). The mean age of the participants in the intervention group and the control group was 40.60±9.37 years (standard deviation) and 40.06±8.61 years respectively, with 72% of the participants being men. The mean number of years of education was similar for both groups. Among the patients in the intervention group, 3 (1%) had early-stage disease (stage I/II) and 65 (46%) had advanced-stage disease (stage III/IV). Among the patients in the control group, 16 (11%) had early-stage disease (stage I/II), and 57 (40%) had advanced-stage disease (stage III/IV). The UICC T, N. and M stages are shown in Table 2. A total of 41 (30%) and 47 (33%) patients received concurrent chemo-radiotherapy in the intervention and control group, respectively, while 25 (18%) patients in each group received induction plus concurrent chemotherapy. Only 5 patients (4%) received targeted therapy in the control group.
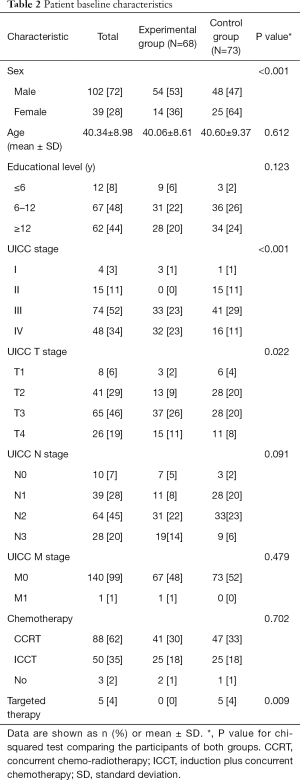
Full table
The CDSM value differences between the groups
Data concerning the CDSM scales and items at pretreatment, immediately after treatment, and 3, 6 and 12 months after IMRT are shown in Tables 3-7.

Full table
Full table

Full table

Full table

Full table
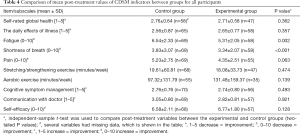
At baseline there were no statistically significant differences between the groups in any CDSM indicators. However, there was a difference in fatigue and shortness of breath between the control and intervention group (6.54±2.33 versus 5.31±2.05, P=0.002; 3.83±3.07 versus 3.34±2.07, respectively, P<0.001) after treatment. There was also a trend towards significance for a difference in pain between the control and intervention group (5.23±2.75 versus 4.35±2.51, respectively, P=0.063).
Significantly increased scores were noted for fatigue (3.78±2.24 versus 3.51±1.80, P=0.025), stretching/strengthening exercise (80.58±99.09 versus 138.53±228.92, P=0.014), and aerobic exercise (369.41±300.46 versus 461.82±451.24, P<0.001) at 3 months after IMRT between the control and intervention group. There was no significant difference between the groups at 6 and 12 months in terms of CDSM indicators (Tables 6,7).
The effect of treatment on CDSM values and its tendency
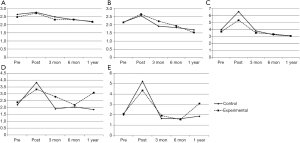
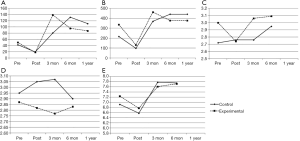
The self-rated global health, the daily effects of illness, fatigue, shortness of breath, and pain indicators demonstrated a reverse checkmark shape (ヘ) while stretching/strengthening exercise, aerobic exercise, cognitive symptom management, and self-efficacy demonstrated a normal checkmark shape (√) in graphs depicting the values at pretreatment, immediately after treatment, and 3, 6, and 12 months post-treatment (Figures 1,2). However, patients in the control group scored higher in terms of communicating with their doctor (Figure 2).
All the data have been uploaded to the research data deposit (RDD) system (RDDA2019001071).
Discussion
Overall, we found statistically significant improvements in NPC participants following an intervention post-treatment in terms of both fatigue and shortness of breath (see Table 4). Participants in the intervention groups also presented with statistically significant improvements at 3 months post-treatment in terms of fatigue, stretching and strengthening exercise, and aerobic exercise (see Table 5). The improvements in fatigue among the intervention participants may be as a result of the intervention itself. A study by Hacker et al. reported that an intervention performed by nurses can potentially reduce fatigue and improve quality of life (16), while another study found that many aspects of the daytime symptomatology of insomnia were improved by nurse-led group treatment (17). Many cancer patients, including NPC patients, lack and need help with basic self-management knowledge and skills. At the same time, cancer is causing escalating health expenditure as the population in 21st century China rapidly ages (18).
The greater improvements in stretching and strengthening exercise and aerobic exercise among intervention participants highlight the importance of nursing interventions. A previous study of our team provides an alternative measure of the comprehensive nutritional index (CNI) to improve the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Head and Neck Cancer Module (EORTC QLQ-H&N35) evaluation system for patients with NPC (14). A cluster randomized controlled trial conducted by Kerse et al. concluded that a nurse counseling intervention improved activity, energy expenditure, health-related quality of life, and hospitalization rates for older primary care patients (19). Nurses often have more contact with patients than doctors, and nurses may play a significant role in psychological and nutritional interventions. NPC patients, particularly at the early stage, typically demonstrate relatively small tumors and include many older adults that can be treated but not cured (20). Furthermore, patients who know more about their disease often fare better than those who rely on IMRT alone. Patients who work to make themselves healthy (for example, by exercising regularly or by losing weight) also fare better (21).
When comparing the post-treatment outcomes of all participants, we found a significant reduction in self-rated global health, the daily effects of illness, fatigue, shortness of breath, and pain, which were mainly attributable to the IMRT, and this tendency gradually improved after treatment (Figure 1). The reverse checkmark shape (ヘ) of the above five indicators when plotted shows that IMRT profoundly affected the patients. In the IMRT epoch, NPC survivors still experience many physical symptoms that affect long-term quality of life many years after treatment, and depression, anxiety, and fatigue remain common in long-term survivors and are highly correlated with quality of life (22,23). There was no significant difference between the two groups except in terms of fatigue and shortness of breath post-treatment. The lack of persistence in the positive effects may be a consequence of lessened statistical power as a result of the sharply decreased sample size at 12 months.
The stretching/strengthening exercise, aerobic exercise, cognitive symptom management, and self-efficacy indicators deteriorated sharply after treatment; this status gradually improved after treatment, and the trend formed a checkmark shape (√) shape when plotted (Figure 2). Once IMRT is initiated, patients do not want to exercise because of fatigue and psychological factors. However, a study by Meneses-Echávez et al. found that supervised physical activity interventions reduce cancer-related fatigue, and the authors suggested that combined aerobic and resistance exercise regimens with or without stretching should be included as part of rehabilitation programs for people who have been diagnosed with cancer (23). The decline in exercise awareness in our participants after treatment highlights the urgent need for health counseling services in clinical practice not only from nurses, but also from other medical staff including doctors. After treatment, the patient’s vitality gradually returned to the pre-admission level or even higher, reflecting enhanced health care awareness after rehabilitation.
Patients in the control group were more positive when communicating with their doctor, but this difference weakened with time (Figure 2). The reason for this may be that the patients had solved most of their problems after consulting the nurse, or it may be that communication with the nurse weakens the role of the doctor. There is currently a huge gap between the need for and the actual delivery of nutritional care services in the clinic (5). Physicians, and especially oncologists, should be aware that nutritional care is much more than enteral or parenteral nutrition, but instead represents a part of a holistic human approach to patient-centered care and is also essential for the successful treatment of patients with cancer (5).
The interpretation of the current study is limited by the fact that many participants did not complete the study, and those data are missing for some outcome variables. Second, there were many confounders, possibly creating a confounding effect. Third, the sample size was relatively small. Nonetheless, the current study demonstrates positive benefits from nutritional counseling for patients with NPC. The current study is the first to document the positive benefits of these programs for NPC patients of Chinese origin. Moreover, it is important to compare the usefulness of the CDSM parameters in an independent validation cohort, ideally collected during a prospective trial. This step is necessary before CDSM can be confidently used to estimate the overall status of NPC patients. Additional research is needed to determine cancer survivors’ unique needs and examine the benefits of tailored versions of the CDSMP. Nevertheless, the CDSMP, at a national and international scale, is a promising intervention and evaluation tool for cancer survivors and should be considered a valuable component of survivorship care.
In conclusion, a nutritional care intervention performed by nurses can greatly reduce fatigue and shortness of breath and greatly increase awareness of stretching/strengthening and aerobic exercise, even 3 months after IMRT treatment in NPC patients.
Acknowledgments
The authors gratefully acknowledge the vital assistance of nurses from the Department of Nasopharyngeal Carcinoma. They completed the questionnaire, helped with data entry, and checked the database. We would also like to thank the head nurse Yuying Fan, who supported this research.
Funding: This study was partly supported by the Medical Research Foundation of Guangdong Province (No. A2014252) and the Nurses Fund of the Sun Yat-sen Cancer Hospital (No. 201301).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/apm-20-1053
Data Sharing Statement: Available at http://dx.doi.org/10.21037/apm-20-1053
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/apm-20-1053). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study protocol was approved by the Research Ethics Committee of SYSUCC, Guangzhou, China (No. B201502401).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Bodenheimer T, Chen E, Bennett HD. Confronting the growing burden of chronic disease: can the U.S. health care workforce do the job? Health Aff (Millwood) 2009;28:64-74. [Crossref] [PubMed]
- Garsa A, Ho JC, Hu C, et al. Bevacizumab is more effective in nasopharyngeal carcinoma patients with lower maximum radiation dose to the temporal lobe. Chin Clin Oncol 2019;8:S20. [Crossref] [PubMed]
- Goeppinger J, Armstrong B, Schwartz T, et al. Self-management education for persons with arthritis: Managing comorbidity and eliminating health disparities. Arthritis Rheum 2007;57:1081-8. [Crossref] [PubMed]
- Erickson N, Paradies K, Buchholz D, et al. Nutrition care of cancer patients-A survey among physicians and nurses. Eur J Cancer Care (Engl) 2018;27:e12855. [Crossref] [PubMed]
- Tomioka M, Braun KL, Compton M, et al. Adapting Stanford's Chronic Disease Self-Management Program to Hawaii's Multicultural Population. The Gerontologist 2012;52:121-32. [Crossref] [PubMed]
- Haslbeck J, Zanoni S, Hartung U, et al. Introducing the chronic disease self-management program in Switzerland and other German-speaking countries: findings of a cross-border adaptation using a multiple-methods approach. BMC Health Serv Res 2015;15:576. [Crossref] [PubMed]
- Smith ML, Wilson MG, Robertson MM, et al. Impact of a Translated Disease Self-Management Program on Employee Health and Productivity: Six-Month Findings from a Randomized Controlled Trial. Int J Environ Res Public Health 2018. [Crossref] [PubMed]
- Smith ML, Towne SD, Herrera-Venson A, et al. Dissemination of Chronic Disease Self-Management Education (CDSME) Programs in the United States: Intervention Delivery by Rurality. Int J Environ Res Public Health 2017. [Crossref] [PubMed]
- Siu AMH, Chan CCH, Poon PKK, et al. Evaluation of the chronic disease self-management program in a Chinese population. Patient Educ Couns 2007;65:42-50. [Crossref] [PubMed]
- Salvatore AL, Ahn S, Jiang L, et al. National study of chronic disease self-management: 6-month and 12-month findings among cancer survivors and non-cancer survivors. Psychooncology 2015;24:1714-22. [Crossref] [PubMed]
- Wagner EH, Ludman EJ, Erin JAB, et al. Nurse Navigators in Early Cancer Care: A Randomized, Controlled Trial. J Clin Oncol 2014;32:12-8. [Crossref] [PubMed]
- Reimer T, Gerber B. Quality-of-life considerations in the treatment of early-stage breast cancer in the elderly. Drugs Aging 2010;27:791-800. [Crossref] [PubMed]
- Zhang LL, Zhou GQ, Qi ZY, et al. Patient- and treatment-related risk factors associated with neck muscle spasm in nasopharyngeal carcinoma patients after intensity-modulated radiotherapy. Bmc Cancer 2017;17:788. [Crossref] [PubMed]
- He Y, Chen L, Chen L, et al. Relationship Between the Comprehensive Nutritional Index and the EORTC QLQ-H&N35 in Nasopharyngeal Carcinoma Patients Treated with Intensity-Modulated Radiation Therapy. Nutr Cancer 2017;69:436-43. [Crossref] [PubMed]
- Ma J, Chen NY, Zhang N, et al. OP0010 Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in patients with locoregionally advanced nasopharyngeal carcinoma: Preliminary results of a phase 3 multicentre randomised controlled trial. Eur J Cancer 2014;50:e3-e4. [Crossref]
- Hacker ED, Peters T, Patel P, et al. Steps to Enhance Early Recovery After Hematopoietic Stem Cell Transplantation: Lessons Learned From a Physical Activity Feasibility Study. Clin Nurse Spec 2018;32:152-62. [Crossref] [PubMed]
- Sandlund C, Hetta J, Nilsson GH, et al. Impact of group treatment for insomnia on daytime symptomatology: Analyses from a randomized controlled trial in primary care. Int J Nurs Stud 2018;85:126-35. [Crossref] [PubMed]
- Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016;66:115-32. [Crossref] [PubMed]
- Kerse N, Elley CR, Robinson E, Arroll B. Is physical activity counseling effective for older people? A cluster randomized, controlled trial in primary care. J Am Geriatr Soc 2005;53:1951-6. [Crossref] [PubMed]
- Chodosh J, Morton SC, Mojica W, et al. Meta-analysis: chronic disease self-management programs for older adults. Ann Intern Med 2005;143:427-38. [Crossref] [PubMed]
- McDowell LJ, Rock K, Xu W, et al. Long-Term Late Toxicity, Quality of Life, and Emotional Distress in Patients With Nasopharyngeal Carcinoma Treated With Intensity Modulated Radiation Therapy. Int J Radiat Oncol Biol Phys 2018;102:340-52. [Crossref] [PubMed]
- Lastrucci L, Bertocci S, Bini V, et al. Late toxicity, evolving radiotherapy techniques, and quality of life in nasopharyngeal carcinoma. Radiol Med 2017;122:303-8. [Crossref] [PubMed]
- Meneses-Echávez JF, Gonzalez-Jimenez E, Ramirez-Velez R. Supervised exercise reduces cancer-related fatigue: a systematic review. J Physiother 2015;61:3-9. [Crossref] [PubMed]


