
Why equianalgesic tables are only part of the answer to equianalgesia
Introduction
Indisputably, opioid therapy will likely be part of the treatment plan for moderate to severe acute pain, cancer pain, and despite current controversy, chronic non-cancer pain. However, opioid therapy is just one part of a treatment plan, and often the therapeutic goal is not achieved. Of course practitioners must consider the presence of pain that is not completely opioid-responsive such as myofascial or neuropathic pain, and select analgesics that better target these types of pain (e.g., gabapentinoids, antidepressants, others). Another strategy may be to switch, or rotate, to a different opioid. There are actually a variety of reasons why a practitioner may need to switch from one opioid to a different opioid, which will be explored in this article. We will also discuss considerations and limitations of using an equianalgesic table to guide opioid switching.
Clinical scenarios that prompt opioid switching
As discussed above, not all types of pain are completely opioid-responsive (lack of effect). Opioid responsiveness has been defined as “the degree of analgesia achieved as the dose is titrated to an endpoint defined either by intolerable side effects or the occurrence of acceptable analgesia.” (1). This clinical phenomenon has been well recognized for many years, and there are several factors described by Mercadante and Portenoy almost 20 years ago (1). Neuropathic pain and often metastatic bone pain may show a variable response to opioid therapy, often requiring a higher than usual opioid dose. Further, we know that patients display variable responsiveness to different opioids (2).
Breakthrough pain, defined as “a transitory exacerbation of pain that occurs on a background of otherwise stable pain in a patient receiving chronic opioid therapy” is a common occurrence in cancer pain (3). This pain may be spontaneous or secondary to an inducing event, which may be volitional or non-volitional. Often breakthrough pain may be of a different etiology than the patient’s persistent pain, or so fleeting (although may be severe) that it is difficult to treat the pain quickly enough, particularly when the breakthrough pain is spontaneous.
Tolerance is defined as “a reduced responsiveness to an opioid agonist such as morphine and is usually manifest by the need to use increasing doses to achieve the desired effect.” (4). One obvious solution to opioid tolerance is to increase the dose of the opioid, continuing to titrate to effect, short of causing adverse effects. Morgan and Christie describe how a 10-fold dosage escalation in chronic pain management is not uncommon, yet paradoxically often a patient is well-controlled on a stable dose for years (4).
When a patient (particularly with a progressive disease) initially responds to opioid therapy, followed by a diminishing response this may be a sign of disease progression. This may be difficult to distinguish from opioid tolerance, but the strategy is similar—increase the opioid dose, titrating to effect, short of causing adverse effects.
As mentioned several times, we titrate opioids to maximal effectiveness, short of causing unacceptable adverse effects. Occasionally the side effect burden becomes too great short of achieving therapeutic success. Switching to a different opioid, which may allow opioid dose reduction, and hopefully fewer adverse effects, is a reasonable strategy.
Other common reasons that necessitate switching to a different opioid regimen include dosage limitations of combination analgesics (e.g., oxycodone/acetaminophen dosing that is approaching or exceeding 4,000 mg acetaminophen per day). Change in a patient’s status often requires switching from one opioid to another. This may be a post-operative patient being switched from a parenteral opioid, now preparing for discharge and continuing oral opioid therapy at home. Or perhaps a patient with an advanced illness who can no longer swallow oral tablets or capsules and now requires transdermal, rectal or parenteral opioid therapy. Patients (or families) often have strongly held opinions about certain opioids (intolerances or allergic responses; stigma) that would be best served by switching opioids. Remaining reasons include opioid shortages, financial reasons (less expensive alternate opioid/dosage formulation), and switching to attempt staying under a mandated daily opioid limit.
Principles of equianalgesia
Switching from one opioid (given by a specific route of administration with a specific dosage formulation) to a different opioid (possibly by a different route of administration and/or dosage formulation) requires knowledge of several opioid-specific variables. These include potency (the intensity of analgesic effect for a given dose), and bioavailability (the percentage of drug that is detected in the systemic circulation after its administration) (1). Potency is determined by how well the opioid accesses the opioid receptor, which is determined by physicochemical (the physical and chemical processes of a drug binding to a receptor) and pharmacokinetic variables (absorption, distribution, metabolism, excretion). These variables can all be determined, therefore construction of an equianalgesic table should be straightforward! Table 1 shows an example of a partial equianalgesic table (5).
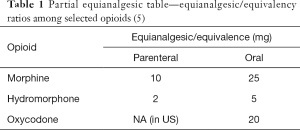
Full table
The concept in using a table such as the example shown are straight forward. An “equianalgesic dose” is defined as “that dose at which two opioids (at steady-state) provide approximately the same pain relief.” (6). For example, 10 mg of parenteral morphine should provide an equivalent amount of analgesic to 25 mg of morphine given by the oral route of administration (at steady state). Another example is 2 mg of intravenous hydromorphone provides approximately equivalent relief to 25 mg of oral morphine. Equianalgesia however, “does not imply, nor was it every intended to assign, an equivalent abuse or respiratory depression risk when comparing two or more opioids.” (7). When we consider the spectrum of variations from patient to patient, it seems obvious that an equianalgesia table has limitations.
Limitations of equianalgesic tables
There are several limitations to an equianalgesia table. First, where did the data come from? The concept of an equianalgesia table is not new, in fact the first table appeared about 50 years ago (8). Most of the studies that informed that table were single-dose studies done in acute post-operative pain and chronic cancer pain. Using opioid-naïve or patients on low-dose opioids (so opioid tolerance was not a high possibility), patients received a high dose and low dose of the study opioid and a high dose and low dose of a comparator opioid (9). Pain reduction was the only outcome assessed, using the sum of pain intensity differences (SPID), and total pain relief (TOTPAR). The results were graphed as a simple, two-point dose-response relationship, and equivalencies were generated (9). We have learned through experience that equivalency data from these single-dose cross-over trials does not always apply to the chronic pain patient. For example, the original equianalgesic table published showed that 10 mg parenteral morphine (intramuscular, intravenous or subcutaneous) was equivalent to 60 mg oral morphine. The single-dose study that led to this recommendation failed to consider the analgesic contribution of the morphine metabolite, morphine-6-glucuronide (10). Subsequent studies have shown the parenteral:oral morphine equivalency at steady-state is closer to 1:2 or 3 (11,12). Data culled from steady-state cross-over trials has emerged in recent years, better informing the equianalgesia chart (13,14)). While the equianalgesic chart has changed somewhat over the years based on newer data, practitioners are slow to change. When asked why, in the face of better data, they are reluctant to change, the answer is often “Because we’ve always done it this way!”
Patient-specific variables are not considered when crafting an equianalgesic table. This includes variables such as age, sex, body surface area, pharmacogenomics (polymorphism of opioid receptors), drug/opioid tolerance (duration and extent of opioid exposure), quantitative and qualitative differences among individuals in cytochrome P450 functions, organ function (liver and kidney), level and stability of pain control, pathogenesis of the pain, drug-drug and drug-food interactions, and comorbid conditions (5,15).
Another concern about equianalgesia tables is the question of bidirectionality. If 10 mg of Drug A gives equivalent pain relief to 20 mg of Drug B, does 20 mg of Drug B give equivalent pain relief to 10 mg of Drug A? Not necessarily! For example, if you look at Table 1, we see that 25 mg oral morphine is approximately equianalgesic to 5 mg oral hydromorphone (a 5:1 ratio). While we assume that the ratio holds in both directions (5:1 morphine:hydromorphone and 1:5 hydromorphone:morphine), but clinical research has shown us when switching from morphine to hydromorphone (including both subcutaneous to subcutaneous, or oral to oral routes), a conversion ratio of 5:1 (morphine:hydromorphone) is seen. However, when switching from morphine to hydromorphone (again, using same route for both opioids) the ratio of 3.7:1 (morphine:hydromorphone) is observed (16).
Several authors have discussed the concept of opioid utility in this issue (17,18). van Dam describes the utility function (where utility = profit – loss) as it applies to pharmacotherapy: utility = benefit – harm (17). Opioid utility is an important consideration in opioid selection. For example, two opioids may provide equivalent pain relief (which is nicely demonstrated in an equianalgesia table) yet one has a higher utility because it causes less harm. Utility is NOT considered or reflected in an equianalgesia table. van Dam further points out “the spectrum of benefit and harm differs, not only among different opioids, but also among different patient populations and pain syndromes.” (17). Might an opioid utility comparative or equivalency table be in our future?
Best practices in opioid switching
Given the limitations of the equianalgesia table, the lack of an opioid utility comparative table, and faced with the need to switch a patient from one opioid regimen to another opioid regimen, what’s a practitioner to do? “Throw your hands in the air and wave ‘em like you just don’t care” (reference to popular culture) seems to be an approach fraught with potential error.
Using a systematic process of patient assessment and calculations is a sound approach. One popular five-step process is as follows (19):
- Step 1—assess the patient’s complaint of pain thoroughly. This allows the practitioner to determine the most likely pathogenesis of the pain, the likelihood that the pain will respond to opioid therapy, and whether this is a new pain complaint, or worsening of a previous pain complaint.
- Step 2—calculate the patient’s total daily use of opioid, including scheduled doses and an average estimation of opioid used to treat breakthrough pain.
- Step 3—use a fair-balance opioid equianalgesic table (such as Table 1) and perform the appropriate ratio calculation, determining the dose of the equivalent, new opioid regimen.
- Step 4—this is an enormous critical thinking step (which online opioid conversion calculators skip entirely)—adjust the calculated dose of the new opioid regimen based on patient-specific information including the assessment garnered in step 1. The practitioner can do one of three things with the newly calculated dose—use that as the dose they select, increase the dose or decrease the dose. If switching from one opioid to a different opioid, it is always prudent to reduce the calculated dose by 25–50% (perhaps less if the switch is being performed due to poorly controlled pain). Note that calculations involving methadone and fentanyl are slightly different and are beyond the scope of this description. One rule of thumb is to be conservative with the scheduled dose of opioid, but generous with the opioid dosed used to treat breakthrough pain (if appropriate to include).
- Step 5—this is probably the most important step of all—monitor the patient carefully in the hours and days that follow the opioid switch, and adjust the regimen based on the patient’s response (therapeutic and potentially toxic).
This five-step process is a safe, and effective approach to switching a patient from one opioid regimen to another opioid regimen, particularly when patient-specific variables are considered, a fair-balance equianalgesia chart is used, and the patient is closely monitored post-switch.
A note of caution about online opioid conversion calculators is warranted in this discussion. There are numerous online calculators available for practitioners to use, and the very nature of all manner of online calculators imbues a sense of confidence in the user. Calculating a creatinine clearance, or risk status based on some numeric system is generally highly accurate, so why wouldn’t users accept an opioid conversion calculator in the same vein? As started above, online opioid conversion calculators are just fancy calculators, basically ONLY performing step 3. They do not inquire about the patient’s clinical status or consider any patient-specific data. It is often quite difficult to even determine which equianalgesic table the application uses, and only a few offer the user the opportunity to dose reduce for lack of complete cross-tolerance. The larger issue is that users tend to “run with” the calculated number and not consider the limitations to equianalgesic tables, and by extension, applications that automate this process. Let the buyer beware, and don’t turn off your brain!
Conclusions
As long as we continue to use opioids to treat acute and chronic pain, there will be a need for skills to switch patients from one opioid regimen to a different opioid regimen. Unfortunately, this is not a cut-and-dried process that is one-size-fits-all. Equianalgesic tables based on the most currently available research are available (5) but these calculations still require careful consideration of the limitations, and patient-specific data. A five-step process for opioid conversions is discussed here, designed to maximize efficacy and minimize toxicity. Perhaps next steps in our equianalgesic journey will be the ability to include consideration of opioid utility.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Mellar P. Davis) for the series “Opioid Utility the Other Half of Equianalgesia” published in Annals of Palliative Medicine. The article was sent for external peer review organized by the Guest Editor and the editorial office.
Conflicts of Interest: The series “Opioid Utility the Other Half of Equianalgesia” was commissioned by the editorial office without any funding or sponsorship. The author has no other conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Mercadante S, Portenoy RK. Opioid poorly-responsive cancer pain. Part 1: Clinical considerations. J Pain Symptom Manage 2001;21:144-50. [Crossref] [PubMed]
- Galer BS, Coyle N, Pasternak GW, et al. Individual variability in the response to different opioids: report of five cases. Pain 1992;49:87-91. [Crossref] [PubMed]
- Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain 1990;41:273-81. [Crossref] [PubMed]
- Morgan MM, Christie MJ. Analysis of opioid efficacy, tolerance, addiction and dependence from cell culture to human. Br J Pharmacol 2011;164:1322-34. [Crossref] [PubMed]
- McPherson ML. Demystifying opioid conversion calculations: A guide for effective dosing, 2nd ed. American Society of Health-System Pharmacists, 2018.
- Shaheen PE, Walsh D, Lasheen W, et al. Opioid equianalgesic tables: are they all equally dangerous? J Pain Symptom Manage 2009;38:409-17. [Crossref] [PubMed]
- Fudin J, Raof M, Wegrzyn EL. Opioid dosing policy: Pharmacological considerations regarding equianalgesic dosing. A white paper from the Academy of Integrative Pain Management. 2017. Available online: Accessed February 2, 2020.https://rsds.org/wp-content/uploads/2014/12/MEDD-White-Paper-FINAL.pdf
- Houde R, Wallenstein S, Beaver W. Evaluation of analgesics in patients with cancer pain. Clin Pharmacy 1966;1:59-97.
- Knotkova H, Fine PG, Portenoy RK. Opioid rotation: The science and the limitations of the equianalgesic dose table. J Pain Symptom Manage 2009;38:426-39. [Crossref] [PubMed]
- Portenoy RK, Thaler HT, Inturrisi CE, et al. The metabolite morphine-6-glucuronide contributes to the analgesia produced by morphine infusion in patients with pain and normal renal function. Clin Pharmacol Ther 1992;51:422-31. [Crossref] [PubMed]
- Kalso E, Valinio A. Morphine and oxycodone hydrochloride in the management of cancer pain. Clin Pharmacol Ther 1990;47:639-46. [Crossref] [PubMed]
- Reddy SK, Agloria M. Pain management. In: The M.D. Anderson Supportive and Palliative Care Handbook. 3rd ed. Houston, TX: The University of Texas MD Anderson Cancer Center; 2008:19-43.
- Reddy A, Yennurajalingam S, Desai H, et al. The opioid rotation ratio of hydrocodone to strong opioids in cancer patients. Oncologist 2014;19:1186-93. [Crossref] [PubMed]
- Reddy A, Vidal M, Stephen S, et al. The conversion ratio from intravenous hydromorphone to oral opioids in cancer patients. J Pain Symptom Manage 2017;54:280-8. [Crossref] [PubMed]
- Smith HS. Variations in opioid responsiveness. Pain Physician 2008;11:237-48. [PubMed]
- Lawlor P, Turner K, Hanson J, et al. Dose ratio between morphine and hydromorphone in patients with cancer pain: a retrospective study. Pain 1997;72:79-85. [Crossref] [PubMed]
- van Dam CJ, Algera MH, Olofsen E, et al. Opioid utility function: methods and implications. Ann Palliat Med 2019. [Epub ahead of print]. [PubMed]
- Olesen AE, Broens S, Olesen SS, et al. A Pragmatic Utility Function to Describe the Risk-Benefit Composite of Opioid and Nonopioid Analgesic Medication. J Pharmacol Exp Ther 2019;371:416-21. [Crossref] [PubMed]
- Gammaitoni AR, Fine P, Alvarez N, et al. Clinical application of opioid equianalgesic data. Clin J Pain 2003;19:286-97. [Crossref] [PubMed]


