
Dynamic evaluation of the correlativity between CD4 cell percentage and FEV1 can predict the balance of the state of the immune system in pulmonary graft versus host disease patients
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is an effective therapy for many hematologic non-malignancies or malignancies (1). In long-term follow-up after transplantation, graft versus host disease (GVHD) remains a major cause of non-relapse mortality. Approximately 50% of SCT recipients develop GVHD. Pulmonary GVHD features irreversible airflow obstruction, which treatment typically involves immunosuppressive regimens. Regardless of whether GVHD caused by immune overreaction or infection caused by immunologic suppression, the end result is death. Therefore, it is crucial to maintain a balance. The CD4 cell percentage can reflect the state of the immune system, whereas FEV1 (% predicted) and FEV1/FVC (actual) can be used to assess the degree of airway obstruction. Here, we present a dynamic evaluation of the correlativity between the CD4 cell percentage and FEV1 that can predict state balance in pulmonary GVHD patients.
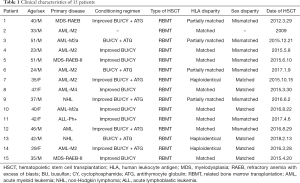
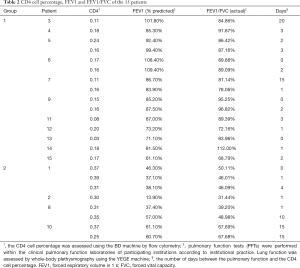
We assessed 15 patients (Table 1 shows clinical characteristics of 15 patients) after HSCT in our hospital and collected CD4 cell percentage, FEV1 and FEV1/FVC data from the same period (Table 2) (The same period was considered less than 20 days between the pulmonary function assessment and the CD4 cell percentage). We plotted these data in scatter plots, which reveal that FEV1 (% predicted) or FEV1/FVC is increased, whereas the CD4 cell percentage is reduced. An analysis of FEV1 (% predicted) and CD4 cell percentage was performed using a bivariate correlation test, and the Pearson correlation coefficient (r) was −0.704 (P=0.000). The relevant FEV1/FVC and CD4 cell percentage values reveal a common conclusion (r=−0.747, P=0.000) Then, we divided the data into two groups according to the CD4 cell percentage. In GROUP 1, CD4 cell percentages were less than 0.25, whereas GROUP 2 included values greater than or equal to 0.25. Comparisons of FEV1 and FEV/FVC between the two groups were both performed using an independent sample t-test. FEV1 in GROUP 1 is increased compared with GROUP 2 (P=0.000). In addition, FEV1/FVC in GROUP 1 is increased compared with FEV1 in GROUP 2 (P=0.000).
Full table
Full table
Pulmonary GVHD features irreversible airflow obstruction and typically occurs within the first 2 years after allo-HSCT. The typical pulmonary function phenotype is characterized as follows: forced expiratory volume in 1 s (FEV1) <70% and FEV1/forced vital capacity (FVC) ratio <0.7. FEV1 and FEV1/FVC are negatively correlated with the severity of pulmonary GVHD. The pathophysiology of GVHD has generally proven to be autoimmune. Furthermore, increasing evidence suggests that GVHD is caused by donor T lymphocytes attacking recipient cells, resulting in immune-mediated tissue damage (2). Numerous experiments have demonstrated that alloreactive T cells move into the thymus and damage medullary thymic epithelial cells (mTECs), resulting in the production of autoreactive CD4 T cells. Autoreactive CD4 T cells are also derived from CD4 T cells in transplants probably through recognizing nonpolymorphic antigens (3-5). Autoreactive T cells interact with donor-derived DCs and B cells, resulting in mutual expansion, autoantibody production and GVHD development (6). During the process, CD4 T cells play an important role, causing GVHD through either thymic-dependent or thymic-independent mechanisms. CD4 cells are mainly involved in humoral immunity with antibody release and represent a potential mechanism leading to inflammation, epithelial destruction, pulmonary fibrosis, and air trapping (7-9). Available evidence indicates that CD4 helper T cells play an important role in the initiation and regulation of GVHD.
Our report demonstrates that the CD4 cell percentage is closely associated with pulmonary function in GVHD patients after HSCT. The results demonstrate that pulmonary function can remain in a better condition when the CD4 cell percentage is less than 0.25. Patients are easily infected when the CD4 cell percentage is extremely low due to excessive immunosuppression. Therefore, lower CD4 cell percentages are not safer. CD4 cell percentages equal to 0.25 offer the most benefits to patients.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cornelissen JJ, Gratwohl A, Schlenk RF, et al. The European LeukemiaNet AML Working Party consensus statement on allogeneic HSCT for patients with AML in remission: an integrated-risk adapted approach. Nat Rev Clin Oncol 2012;9:579-90. [Crossref] [PubMed]
- Grønningsæter IS, Tsykunova G, Lilleeng K, et al. Bronchiolitis obliterans syndrome in adults after allogeneic stem cell transplantation-pathophysiology, diagnostics and treatment. Expert Rev Clin Immunol 2017;13:553-69. [Crossref] [PubMed]
- Zhao D, Young JS, Chen YH, et al. Alloimmune Response Results in Expansion of Autoreactive Donor CD4 T Cells in Transplants That Can Mediate Chronic Graft-versus-Host Disease. J Immunol 2011;186:856-68. [Crossref] [PubMed]
- Rangarajan H, Yassai M, Subramanian H, et al. Emergence of T cells that recognize nonpolymorphic antigens during graft-versus-host disease. Blood 2012;119:6354-64. [Crossref] [PubMed]
- Young JS, Wu T, Chen Y, et al. Donor B Cells in Transplants Augment Clonal Expansion and Survival of Pathogenic CD4 T Cells That Mediate Autoimmune-like Chronic Graft-versus-Host Disease. J Immunol 2012;189:222-33. [Crossref] [PubMed]
- Wu T, Young JS, Johnston H, et al. Thymic Damage, Impaired Negative Selection, and Development of Chronic Graft-versus-Host Disease Caused by Donor CD4 and CD8 T Cells. J Immunol 2013;191:488-99. [Crossref] [PubMed]
- Hostettler KE, Halter JP, Gerull S, et al. Calcineurin inhibitors in bronchiolitis obliterans syndrome following stem cell transplantation. Eur Respir J 2014;43:221-32. [Crossref] [PubMed]
- Fukami N, Ramachandran S, Saini D, et al. Antibodies to MHC Class I Induce Autoimmunity: Role in the Pathogenesis of Chronic Rejection. J Immunol 2009;182:309-18. [Crossref] [PubMed]
- Williams KM, Chien JW, Gladwin MT, et al. Bronchiolitis Obliterans After Allogeneic Hematopoietic Stem Cell Transplantation. JAMA 2009;302:306-14. [Crossref] [PubMed]


