Communicating prognosis of patients with advanced cancer between health care providers: a tertiary cancer center review of written correspondence
Introduction
Informing patients with advanced cancer of their likely prognosis is a balance of engendering hope and providing honest disclosure (1). Discussions of prognosis between oncologists and their patients enable early goal setting and treatment planning, not only for the patient but also for their family and carers. Despite oncologists self-reporting frequent discussion of disease incurability, previous studies highlight that a quantitative prognosis is not usually provided to patients (2,3) and this contrast to an overall high level of information need identified by patients with life-limiting diseases and their caregivers in English-speaking countries (4).
Documentation of such discussions are crucial for other health care providers (HCPs) in the provision of holistic care during the terminal stage of their patients’ illness. In the community, family physicians have a key a role in their patients’ care due to their accessibility, often long-standing rapport with patients and their families, and ability to coordinate healthcare resources (5). For hospital-based HCPs, while some may be actively involved in patient treatment, others may not be familiar with the patient. This is particularly relevant for patients presenting to the emergency department for whom the first physician they meet will most likely be unaware of the patient’s prognosis. Similarly, hospital intensivists and other admitting physicians will face barriers in providing patient-centred care where there is a lack of awareness of prognosis and goals-of-care (GOC). Thus, the communication of prognosis by the oncologist to other HCPs may influence whether appropriate care is provided according to the patient’s likely outcome and preferences, particularly in the evolving landscape of treatment options available for advanced cancer, although it remains unclear if life expectancy information unequivocally leads to better health outcomes.
Although verbal communication between HCPs may occur, outpatient (OP) letters and discharge summaries following a hospital admission are key forms of written communication. In hospital, HCPs refer to previous internal consultation notes and discharge summaries to gather an understanding of the patient’s prognosis. When communicating prognosis, the documentation can be broadly categorized into qualitative (e.g., “palliative goal of therapy”) or quantitative (e.g., “typical survival <12 months”) terms.
The nature, frequency and timing of prognostic information documented in these various forms of written correspondence between HCPs is currently unknown. We therefore evaluated the documentation of prognosis in such written correspondence by oncologists in an Australian tertiary centre with the overall aim of determining how effectively patient prognosis is communicated to other treating clinicians.
Methods
This retrospective, medical record review study included consecutive patients aged ≥18 years with incurable solid organ cancer who were admitted and subsequently died under the medical oncology or palliative care unit at a large, tertiary public hospital from January 1 to December 31, 2015. At least 1 encounter in medical oncology OP clinic within the patient’s last year of life was required for study inclusion. At the study center, patients undergoing palliative treatment for incurable cancer were not mandatorily referred to hospital-based or community-based palliative care services. We excluded patients actively enrolled in a clinical trial due to a lack of electronic documentation for review on these patients, and patients being treated with curative intent. The study was approved by the institutional ethics review board of Austin Health (LNR/16/Austin/212).
Data were collected regarding patient baseline demographics, cancer diagnosis and date of death. In relation to OP clinical encounters, the total number and date of OP oncology clinic consultations within the last year of life were collected. For inpatient encounters prior to patients’ final admission and subsequent death, we obtained the total number and date of inpatient admissions under the medical oncology unit within the last year of life.
Correspondence data were collected and categorized as either external or internal. External correspondence was defined as any patient information that was sent to members of the patient’s treating team who were not directly affiliated with the hospital, such as family physicians and other medical specialists. This was in the form of OP letters from the medical oncology specialist following oncology clinic visits, or discharge summaries following inpatient medical oncology admissions. Discharge summaries are typically completed by junior medical staff and addressed to community HCPs as an official summary of the admission, including changes in care and record of family meetings. Internal correspondence was defined as any patient information that was only accessible to hospital staff and included any consultation notes by medical oncology specialists from OP visits.
We examined correspondence data for any documentation of patient prognosis by categorizing prognosis as either qualitative or quantitative (Table 1). Statements of qualitative prognosis were considered a broad discussion of the patient’s overall clinical disposition and/or goals of treatment. We excluded statements of treatment description alone (e.g., “chemotherapy”), unless it referred to the treatment aim (e.g., “palliative chemotherapy”) and excluded statements that referred to patients’ clinical progress alone (e.g., “not responding to treatment”), as this was deemed to not adequately convey overall disposition. Quantitative prognosis was defined as an estimate of the patient’s prognosis with an inferred timeframe (e.g., “months” or range “2–6 months”). The earliest date of qualitative and quantitative prognosis documentation was used to determine the timing of communication in relation to patient death. In the event that only a statement of quantitative prognosis was documented according to our definition, this was also counted towards a documented qualitative prognosis. All prognostic documentations were independently evaluated by three study investigators (Anis A. Hamid, Francis J. Ha, Oindrila Das) according to specific criteria determined a priori and verified by the senior author (Andrew J. Weickhardt) in the case of any discrepancy.

Full table
Data relating to documentation of GOC and an advance care plan (ACP) were also collected. GOC refers to limitations in medical treatment in situations of acute deterioration such as suitability for cardiopulmonary resuscitation, intensive care support or intubation, and is designed as a replacement for not-for-resuscitation orders (6). GOC forms outlining these treatment goals are completed by medical staff in discussion with the patient and family, upon each admission to medical inpatient services and is a requirement for admission to inpatient palliative care. GOC forms reflect discrete medical care goals related to the associated hospital admission and are not included in correspondence to community HCPs, therefore they were not reviewed for prognostic content. An ACP refers to legal documentation outlining the patient’s preferences for medical treatment and limitations of interventions at the end of life (7) and does not contain prognostic information. An ACP is typically completed in the OP setting by a dedicated ACP team at our center and is associated with his or her electronic medical record.
We used descriptive statistical analysis with categorical variables presented as numerical value with percentages and continuous variables presented as medians with their associated interquartile range (IQR).
Results
Patients
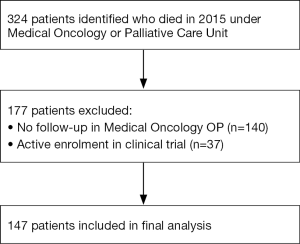
Three hundred and twenty-four patients were admitted and died during inpatient palliative care or medical oncology admission during the study period. A total of 177 were excluded from analysis primarily because of lack of OP medical oncology follow-up at the study center (n=140) and clinical trial enrolment (n=37) (Figure 1). One hundred and forty-seven patients were included in the final analysis. The median age of patients at time of death was 70 (IQR, 58–77) years with various common organ malignancies represented (Table 2).

Full table
Hospital-based care encounters
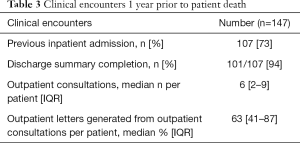
Patients attended a median of 6 (IQR, 2–9) medical oncology OP consultations in the last year of life. Most (107 patients, 73%) had an inpatient hospital admission during this time, with first admission occurring at a median of 4.7 months (IQR, 2.0–8.2 months) prior to death.
Documentation and communication of prognosis
Of the eligible patients comprising the analysis cohort, a total of 881 OP consultation notes, 544 OP letters and 243 discharge summaries were reviewed. The median proportion of medical oncology OP consultations per patient resulting in an OP letter was about two-thirds (median 63%, IQR, 41–87%). The rate of discharge summary completion for inpatient admissions was high (94%). Table 3 summarizes clinical encounters and associated correspondence to external providers.
Full table
OP consultation notes contained qualitative prognosis in 63% of patients with a similar proportion observed for corresponding OP letters (61%). Twenty-four patients (26%) with qualitative prognosis documented in OP consultation notes did not have qualitative prognosis document in an OP letter. When qualitative prognosis was documented in OP letters, this occurred at a median of 3.5 months (IQR, 1.6–6.9 months) prior to death. 50% of all discharge summaries included any documentation of qualitative prognosis (Figure 2).
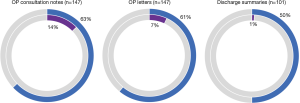
The frequency of documented quantitative prognosis was markedly lower across internal [OP consultation notes: 14% of patients, median 3.4 months (IQR, 0.7–4.4 months) before death] and external correspondence [OP letters: 7% of patients, median 2.2 months (IQR, 1.1–4.4 months) before death] (Figure 2). All patients with quantitative prognosis documentation had concurrent documentation of qualitative prognosis present. One discharge summary (1%) documented quantitative prognosis.
Almost all patients had documentation of GOC (99%) however only 15% had a completed ACP accessible to the hospital treating team and this was performed at a median of 3.7 months (IQR, 1.3–7.7 months) prior to death.
Discussion
We conducted a retrospective study of prognosis documentation in patients with incurable malignancies during their last year of life. Our main findings are that quantitative information about patient prognosis is infrequently communicated in medical oncology documents. When completed, first correspondence of prognostic information typically occurred within the 6-month period prior to a patient’s death. These results highlight that prognosis is poorly documented and likely reflects the lack of communication between oncologists and other HCPs which could have implications in delivering patient-centred care.
While the nature of verbal communication of prognosis to patients has been extensively reviewed (8-10), the communication of prognosis between primary oncologists and other HCPs is less studied (11). In our study cohort, no legal standard of prognosis documentation exists. Only recently has the American Society of Clinical Oncology established a consensus guideline addressing patient-clinician communication and documentation of prognosis, goals of care and end-of-life directives (12) which highlights documentation of GOC discussions as a core recommendation. The open disclosure of prognosis of cancer patients to other HCP is essential to allow appropriate treatment for concurrent and perhaps emergent medical issues, and to provide additional informed team members to counsel patients faced with an incurable, poor prognosis disease. Arguably provision of this documentation may avoid issues concerning over-estimating prognosis by other HCPs in discordance to the treating oncologist (13), and adopting inappropriate life-extending and aggressive treatments that may be given by other uninformed HCPs (14). Conversely, in the era of novel cancer therapies where patients can achieve a durable tumour response and significantly improved quality of life, it is important to inform other non-oncology specialists that the prognosis for some patients has improved, and more aggressive treatments may be reasonable. Unless documented in external or internal correspondence, this information is not readily available in emergent situations to other HCPs.
Given oncologists’ estimations of life expectancy improves with declining patient performance status (15,16) we decided to only evaluate documented prognostic information in the 12 months prior to death, hypothesising that there may be an increased tendency of oncologists to convey this information within this timeframe. We observed that, when a quantitative prognosis was documented (only 7% of patients in OP letters; 14% of patients in internal notes), it occurred close to death which could reflect greater certainty from the treating oncologist. Studying oncologists’ attitudes towards discussing prognosis reveal recurring themes—fear of making prognostic errors, concern of losing patient confidence, statistical information ‘robbing’ patients of hope and a preference for open communication over ‘numbers’ (8-10). Indeed, these may also present similar barriers to documenting prognostic information to other HCPs. Predictions of life expectancy can be imprecise but well-calibrated (17), allowing for practical application of ‘worse-case’, ‘typical’ and ‘best-case’ scenarios using multiples of median survival and incorporation of knowledge from contemporary clinical trials of similar patients. This has been demonstrated as a feasible approach in patient cohorts receiving first-line therapy for incurable breast and lung cancer (18-20).
GOC documentation was completed in almost all patients (99%) in the 12 months prior to death, reflecting a hospital requirement for all inpatient admissions. GOC should be created by clinicians with patients and caregivers, and should utilize pre-existing advanced care planning documentation. An ACP reflects patients’ values and beliefs, considers future situations they may find unacceptable or too burdensome in relation to their health, states specific treatments that the patients would not want considered, and has scope to nominate and provide directions for substitute decision makers. While an ACP is supported for all patients (21), there are barriers to implementing this policy reflected in the low proportion of patients (15%) with an ACP in place. Those barriers include time for clinicians to create an ACP with a patient, lack of trained staff, perceived lack of time to create an ACP within a busy hospital environment and a perceived lack of utility regarding an ACP relative to a more treatment-focused GOC. The results of clinical trials assessing the utility and impact of ACP on outcomes of patients with cancer are eagerly awaited, and may impact uptake (22).
There are several limitations that should be acknowledged in this study. First, we examined only written forms of communication and it is uncertain whether any verbal discussions or other unaudited forms of communication (for example, referral to community services) containing prognostic information may have occurred between oncologists and other HCPs. Second, the generalizability of the results is dependent on the model of care and patient population of other health services. In Australia, models of OP oncology care vary between public hospitals. For example, some oncology departments may employ an OP model where a patient is assigned to a specific provider and managed by them solely as the primary oncology physician (similar to a private care setting). In contrast, our institution usually adopts a model of mixed medical oncology providers (both junior and senior) at OP review, dependent solely on next availability. While this has increased the number of oncology providers contributing to correspondence in this study, it may result in inter-provider variation in patient familiarity, awareness of previous prognostic discussions and quality of documentation. Furthermore, documentation patterns may differ between patients who are more likely to die during hospital admission compared to patients who die in the community. Our data may not reflect health care settings with differences in institution-specific guidelines and/or health policy and legislation-level documentation requirements.
Conclusions
We observed that prognosis, particularly quantitative information, is infrequently documented in regular correspondence to external HCPs despite the poor prognosis of the patient cohort. In their last 12 months of life, most patients would be expected to demonstrate clear physical, functional and radiological signs of progressive disease and deterioration over this period. Documentation of these changes and the prognostic implications they carry is pertinent to convey to other members of the treating team in both internal and external correspondence. There remains an unmet need to improve communication and documentation of prognosis in order to promote cohesive care between hospital and community providers.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the institutional ethics review board of Austin Health (LNR/16/Austin/212).
References
- Kodish E, Post SG. Oncology and hope. J Clin Oncol 1995;13:1817. [Crossref] [PubMed]
- Daugherty CK, Hlubocky FJ. What are terminally ill cancer patients told about their expected deaths? A study of cancer physicians' self-reports of prognosis disclosure. J Clin Oncol 2008;26:5988-93. [Crossref] [PubMed]
- Gattellari M, Voigt KJ, Butow PN, et al. When the treatment goal is not cure: are cancer patients equipped to make informed decisions? J Clin Oncol 2002;20:503-13. [Crossref] [PubMed]
- Parker SM, Clayton JM, Hancock K, et al. A systematic review of prognostic/end-of-life communication with adults in the advanced stages of a life-limiting illness: patient/caregiver preferences for the content, style, and timing of information. J Pain Symptom Manage 2007;34:81-93. [Crossref] [PubMed]
- Ramanayake RP, Dilanka GV, Premasiri LW. Palliative care; role of family physicians. J Family Med Prim Care 2016;5:234-7. [Crossref] [PubMed]
- Thomas RL, Zubair MY, Hayes B, et al. Goals of care: a clinical framework for limitation of medical treatment. Med J Aust 2014;201:452-5. [Crossref] [PubMed]
- Silverman HJ, Vinicky JK, Gasner MR. Advance directives: implications for critical care. Crit Care Med 1992;20:1027-31. [Crossref] [PubMed]
- Christakis NA, Iwashyna TJ. Attitude and self-reported practice regarding prognostication in a national sample of internists. Arch Intern Med 1998;158:2389-95. [Crossref] [PubMed]
- Rogg L, Aasland OG, Graugaard PK, et al. Direct communication, the unquestionable ideal? Oncologists' accounts of communication of bleak prognoses. Psychooncology 2010;19:1221-8. [Crossref] [PubMed]
- Butow PN, Dowsett S, Hagerty R, et al. Communicating prognosis to patients with metastatic disease: what do they really want to know? Support Care Cancer 2002;10:161-8. [Crossref] [PubMed]
- Moth EB, Parry J, Stockler MR, et al. Doctor-to-doctor communication of prognosis in metastatic cancer: a review of letters from medical oncologists to referring doctors. Intern Med J 2015;45:909-15. [Crossref] [PubMed]
- Gilligan T, Coyle N, Frankel RM, et al. Patient-Clinician Communication: American Society of Clinical Oncology Consensus Guideline. J Clin Oncol 2017;35:3618-32. [Crossref] [PubMed]
- Gramling R, Fiscella K, Xing G, et al. Determinants of Patient-Oncologist Prognostic Discordance in Advanced Cancer. JAMA Oncol 2016;2:1421-6. [PubMed]
- Weeks JC, Cook EF, O'Day SJ, et al. Relationship between cancer patients' predictions of prognosis and their treatment preferences. JAMA 1998;279:1709-14. [Crossref] [PubMed]
- Glare P, Virik K, Jones M, et al. A systematic review of physicians' survival predictions in terminally ill cancer patients. BMJ 2003;327:195-8. [Crossref] [PubMed]
- Lambden J, Zhang B, Friedlander R, et al. Accuracy of Oncologists' Life-Expectancy Estimates Recalled by Their Advanced Cancer Patients: Correlates and Outcomes. J Palliat Med 2016;19:1296-303. [Crossref] [PubMed]
- Stockler MR, Tattersall MH, Boyer MJ, et al. Disarming the guarded prognosis: predicting survival in newly referred patients with incurable cancer. Br J Cancer 2006;94:208-12. [Crossref] [PubMed]
- Kiely BE, Alam M, Blinman P, et al. Estimating typical, best-case and worst-case life expectancy scenarios for patients starting chemotherapy for advanced non-small-cell lung cancer: a systematic review of contemporary randomized trials. Lung Cancer 2012;77:537-44. [Crossref] [PubMed]
- Kiely BE, Martin AJ, Tattersall MH, et al. The median informs the message: accuracy of individualized scenarios for survival time based on oncologists' estimates. J Clin Oncol 2013;31:3565-71. [Crossref] [PubMed]
- Kiely BE, Soon YY, Tattersall MH, et al. How long have I got? Estimating typical, best-case, and worst-case scenarios for patients starting first-line chemotherapy for metastatic breast cancer: a systematic review of recent randomized trials. J Clin Oncol 2011;29:456-63. [Crossref] [PubMed]
- Palliative Care Australia: Advanced Care Planning - Position Statement. Available online: www.palliativecare.org.au
- Detering KM, Hancock AD, Reade MC, et al. The impact of advance care planning on end of life care in elderly patients: randomised controlled trial. BMJ 2010;340:c1345. [Crossref] [PubMed]