Radiotherapy for patients with unresected locally advanced breast cancer
Introduction
Locally advanced breast cancer (LABC) cases constitute 5–10% of newly diagnosed breast cancers in North America (1). LABC includes patients with tumours >5 cm, involvement of the chest wall, muscle, or skin, satellite nodules, or extensive nodal involvement (2,3). Postoperative radiotherapy has been shown to improve locoregional control and overall survival (OS) in patients with LABC (4,5).
In patients without distant metastatic disease, neoadjuvant radiotherapy is usually reserved for cases where the tumour is unresectable following neoadjuvant systemic therapy. In such cases, RT may be given with the aim of rendering the tumour operable and allowing for curative surgery.
For LABC patients with synchronous distant metastases, the utility of local treatments such as surgery and/or RT to the primary tumour is controversial (6-8); one randomized study demonstrated no difference in OS although locoregional control was improved (6). Locoregional treatment may, however, be offered to LABC patients with distant metastasis for the purpose of palliating symptoms. In these cases RT is generally preferred to surgery, as it is less invasive and does not require general anaesthetic (8). The purpose of this study was to evaluate the effectiveness of RT for local control in patients with unresected LABC, as measured by LPFS and when appropriate, successful subsequent surgery. Patients were grouped into those who had no evidence of distant disease at the time of radiotherapy (group 1) and those who did (group 2); OS and DPFS were also evaluated.
Methods
A retrospective review of patients with LABC treated with external beam RT between 2004 and 2016 at Sunnybrook Health Sciences Centre was conducted with institutional research ethics board approval. Patients with pathologically confirmed triple negative breast cancer were excluded from this analysis, since it is a distinct phenotype associated with poor prognosis and relative radioresistance (9). Patients receiving low-dose palliative regimens with a 2 Gy per fraction equivalent dose (EQD2) of less than 40 Gy (i.e., 8 Gy/1, 20 Gy/5, and 30 Gy/10 fractionation schedules) were excluded to maintain consistency within the study population.
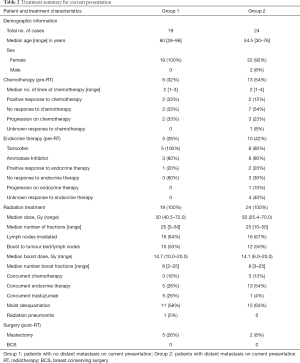
Demographic information including age and sex was collected. Treatment information included date of diagnosis, tumour size, disease stage, hormone receptor and HER-2 status, systemic therapy treatments, and RT dose, fractionation and technique. Where the current presentation was a recurrence, treatment information such as surgery, locoregional RT, and systemic therapy from the previous diagnosis was obtained where possible. Patient-reported symptoms and side effects of RT as documented by healthcare professionals were recorded. Patients were stratified based on the presence of metastatic disease at the time of radiotherapy.
Patients without distant metastatic disease at presentation were included in group 1, and patients with distant metastases at presentation were designated as group 2. The locations of distant metastases were reported where applicable.
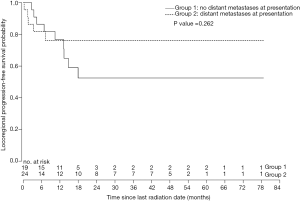
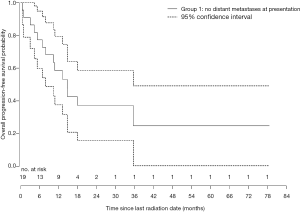
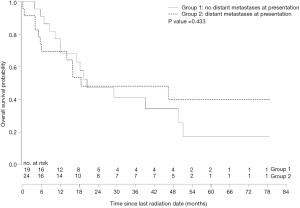
The primary endpoint for patients in group 1 was successful surgical excision of the primary tumour with clear margins. Secondary endpoints were LPFS, radiological and clinical response to RT, distant progression-free survival (DPFS), overall progression-free survival (OPFS), and OS. For patients in group 2, the primary endpoint was LPFS, and secondary endpoints were radiological and clinical response to RT, and OS. Because cause of death was unknown for several patients, disease-specific survival was not calculated. OPFS for patients in group 1 was calculated using the Kaplan-Meier method. OS and LPFS were plotted for both groups using the Kaplan-Meier method, and compared using the log-rank test.
Patients’ response to RT was determined using radiological imaging, clinical measurements, and clinical notes. Baseline tumour size was measured from the computed tomography (CT) radiation planning scans which were consistently done just prior to starting RT. Imaging reports completed after RT were reviewed and the tumour response to RT was assessed based on the notes and impression reported by the radiologist reviewing the imaging. Where imaging was unavailable, RT response was determined using clinical notes dictated by the patient’s oncologists. Data regarding symptoms and adverse events were obtained from clinical notes.
A complete response was defined when the index lesion was no longer identifiable. Partial response was a decrease in size of the index lesion relative to baseline, and stable response was defined as gross stability of the index lesion. Progression referred to an increase in size of the index lesion relative to baseline. Locoregional progression was any recurrence or progression in the ipsilateral breast, chest wall, axillary, inframammary, infraclavicular, or supraclavicular lymph nodes, or skin within the treatment field. Progression or metastasis in any other area was considered distant progression.
OS was defined as the time from the end of RT to death or censored at the most recent follow-up. LPFS and DPFS were defined as the time from the end of RT to the first diagnosis of locoregional and distant progression, respectively. Overall progression-free survival (OPFS) was the time from the end of RT to the first diagnosis of either locoregional or distant progression. For patients with no locoregional or distant progression, LPFS and DPFS were calculated as the time from RT completion to death or last follow-up. OS, LPFS, and OPFS estimates were also calculated using the Kaplan-Meier method, to account for the patients who were lost to follow-up before an event (progression or death) occurred. All calculations were conducted using Statistical Analysis Software (SAS version 9.4 for Windows), and P values of <0.05 were considered statistically significant.
Results
There were 42 patients and 43 treatment sites included in the present analysis, as one patient was treated for bilateral LABC. Forty-five patients had pathologically-confirmed triple-negative breast cancer; these were excluded from the present study. Patient and disease characteristics are summarized in Table 1. Forty-one cases (95%) occurred in women and 2 (5%) in men. In 24 cases (56%) there was known distant metastatic disease at presentation, and in the remaining 19 (44%) there was not. The median ages were 60 years (range, 28–96 years) and 54.5 years (range, 30–76 years) in groups 1 and 2, respectively.

Full table
Tumour and treatment characteristics
Of the 43 cases evaluated in this study, 3 tumours (7%) were stage T2, 4 (9%) were T3, 34 (79%) were T4, and 2 (5%) were unknown. Four cases (9%) were N0, 18 (42%) were N1, 5 (12%) were N2, 14 (33%) were N3, and 2 (5%) were unknown. There was known lymphovascular invasion in 38 (88%) of the cases, and skin involvement in 34 (79%). Most cases (25, 58%) were ER/PR positive and HER-2 negative. Nine tumours (21%) were positive for HER-2 overexpression. Fourteen cases (33%) were recurrences; all of these patients had surgery on first presentation and 5 had RT on first presentation (Table 1). Patients experienced ulceration or bleeding in 24 cases (56%) and breast/chest wall pain in 9 cases (21%). Among the 24 patients with known metastatic disease at presentation (group 2), 63% had bone metastases and 54% had visceral (lung, brain, or liver) metastases (Table 1).
The median follow-up time was 14.3 months (range, 0–79.9 months) from the end of RT for the entire cohort, 15.9 months (range, 0–79.8 months) in group 1 and 13.7 months (range, 0–79.9 months) in group 2. The median follow-up from diagnosis was 25.0 months (range, 2.5–158.2 months) for the entire cohort, 23.2 months (range, 2.5–158.2 months) in group 1 and 30.1 months (range, 3.0–87.7 months) in group 2.
At least one cycle of chemotherapy was administered prior to RT in 19 cases (44%). All patients received external beam RT to the breast or chest wall, and regional lymph nodes were treated in 32 cases (74%). The median RT dose delivered to the breast or chest wall was 50 Gray (Gy) (range, 25.4–72 Gy) in 25 fractions (range, 5–50 fractions) for both groups. One patient was prescribed 65 Gy in 50 twice-daily fractions, but due to systemic disease progression was switched to a shorter regimen. This patient received 25.4 Gy in 11 fractions altogether, and was included in this study based on the original prescribed dose of 65 Gy. Twenty-one cases (49%) received an additional boost to the tumour bed or regional lymph nodes (Table 2). Chemotherapy and hormonal therapy were administered concurrently with RT in 6 (14%) and 18 (42%) of cases, respectively (Table 2). Twenty-eight patients (65%) received hormonal therapy and 22 (51%) were treated with chemotherapy either before, during, or after RT.
Full table
Tumour response
In most cases (30, 70%) the maximal response to RT occurred within 3 months of completing RT. Three months after RT completion, 7 cases (16%) demonstrated a complete response, 29 (67%) showed a partial response, 1 (2%) remained stable, and 5 (12%) experienced locoregional progression. One patient was not evaluated within 3 months of RT but within 6 months demonstrated a complete response. Over the entire follow-up period, 13 cases (30%) had locoregional progression: 8 (42%) in group 1 and 5 (21%) in group 2 (Table 3).
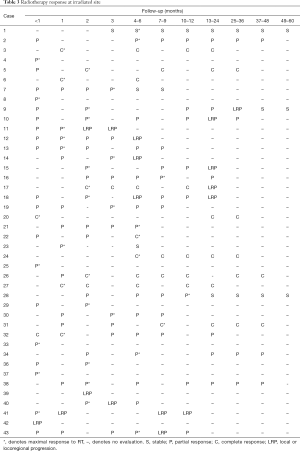
Full table
In 24 cases (56%) the tumours were ulcerating or bleeding. After completion of RT, decreased ulceration or bleeding was noted in 13 of these cases (54%).
Treatment toxicity
Skin toxicity was documented by healthcare professionals. Moist desquamation occurred in 60% of cases, but no patients experienced grade 4 or 5 radiation dermatitis. One patient developed decreased breath sounds, crackles, and a non-productive cough consistent with clinical and radiological (CT scan) evidence of radiation pneumonitis. The patient’s symptoms were treated with prednisone.
Progression-free survival
Five of the 19 patients in group 1 (26%) underwent mastectomies with negative margins after completing RT. The median LPFS was 13.4 months (range, 0–79.8 months) for patients in group 1. Patients in group 1 who had the primary tumour removed after RT had a median LPFS of 6.9 months (range, 2.8–79.8 months), versus those who did not (median 14.3 months; range, 0–62.4 months). In group 2, the median LPFS was 8.2 months (range, 0–79.9 months) overall. Two patients in group 2 had mastectomy following RT and had a median LPFS of 66.5 months (range, 53.2–79.9 months). LPFS among the remaining patients in group 2, who did not have surgery, was 6.70 months (range, 0–56.0 months). LPFS was not significantly different between patients in groups 1 and 2 (P=0.262; Figure 1).
The median DPFS for patients in group 1 was 10.6 months (range, 0–79.8 months). Among patients who had surgery following RT, the median DPFS was 11.7 months (range, 0.9–79.9 months). The median DPFS of the remaining patients was 8.0 months (range, 0–51.9 months).
The median OPFS for patients in group 1 was 11.7 months (range, 0–79.8 months) (Figure 2). The median OPFS was 11.7 months (range, 0.93–79.8 months) in patients who had surgery after RT, and 10.7 months (range, 0–29.5 months) in patients who did not. Since DPFS was not calculated for patients in group 2, OPFS for these patients was not recorded.
OS
Twenty-six patients died before this review. For the remaining 17 patients, OS was calculated until the patient’s most recent follow-up at our institution. One patient in group 1 and one patient in group 2 did not return for any follow-up appointments following completion of RT.
Patients in group 1 had a median OS of 15.9 months (range, 0–79.8 months). The five patients who had mastectomy after completing RT had a median OS of 12.1 months (range, 3.9–79.8 months). Among patients who did not have surgery, the median OS was 16.2 months (range, 0–62.4 months).
The median OS in group 2 was 13.7 months (range, 0–79.9 months). In the patients who had mastectomy after completion of RT, the median OS was 66.5 months (range, 53.2–79.9 months) compared to 11.4 months (range, 0–56.0 months) in the patients who did. OS was not significantly different from group 1 (P=0.433; Figure 3).
Discussion
Patients in this study presented with very advanced breast cancer: 79% had T4 disease, 33% had N3, and the majority experienced primary tumour symptoms such as pain, bleeding or ulceration. Most patients had prior treatment with hormonal therapy and on average, two different lines of chemotherapy prior to RT. RT was used in these cases to gain local control of patients’ disease, either to achieve enough tumour shrinkage such that surgical resection was possible, or to palliate symptoms.
To our knowledge, there are no prospective studies describing the clinical efficacy of RT alone for improving resection or for palliation of symptomatic LABC. Three prior studies have examined the use of concurrent chemoradiation for unresectable breast tumours after failing first line neoadjuvant chemotherapy (10-12). Kao et al., reported that 13 of 16 patients (81%) with unresectable disease treated with vinorelbine and RT proceeded to mastectomy (10). A study of concurrent capecitabine and radiotherapy for anthracycline resistant disease by Gaui et al. reported that 23 of 28 patients (82%) were rendered operable following concurrent chemoradiation (11). Kosma et al. described a cohort of 17 patients with ‘far advanced’ LABC treated with concurrent fluorouracil and radiotherapy after progression on chemotherapy where 3 patients (18%) proceeded to mastectomy (12). For group 1 in the current study, only 5 of 22 patients (22%) had surgery after completion of RT. Surgical resection was presumed to be the goal of RT in this group and, by making curative treatment possible, RT might have been expected to significantly improve OS; however, some of these patients may actually have been treated with very little likelihood of subsequent surgery, due to the tumour extent or medical co-morbidities.
The optimal locoregional treatment approach for patients presenting with metastatic breast cancer remains controversial, as are the benefits of surgery and/or radiotherapy in this group (6-8). Badwe et al. completed a randomized study of surgery and RT following systemic therapy for patients with metastatic breast cancer, compared with no upfront locoregional treatments, and found no difference in OS between groups (6). Patients who underwent surgery and RT had significant improvement in LPFS but interestingly, poorer distant metastasis-free survival. In contrast, an institutional review of 581 patients presenting with stage 4 breast cancer by Le Scodan et al. demonstrated an improvement in OS in those who had locoregional therapy (most commonly RT without surgery) compared with those who did not (8); however, the potential for selection bias should be noted, as patients with better prognoses likely received more aggressive treatment, such as surgery in addition to RT. Bourgier et al. also examined the use of locoregional treatments in patients with metastatic breast cancer within a large institutional series (13). Patients treated with surgery plus RT had no improvements in OS or progression free survival compared with patients receiving RT only. Additional randomized studies of surgery versus no surgery are currently underway, which might identify specific indications for surgery in the setting of metastatic disease (7). At our institution, patients with metastatic disease usually undergo systemic therapy; RT is reserved for progression or locoregional symptom management. Surgery is only considered in exceptional cases where a long disease-free interval might be expected. Because these patients usually undergo RT with palliative intent, quality of life and symptom burden are the primary outcome measures, and less emphasis is placed on the effect of RT on OS. In group 2 of the current study, two patients had surgery after completion of RT. Both had pulmonary nodules at presentation, and were offered surgery after those nodules had been monitored for several months after systemic therapy and remained stable. The median OS of the two patients was 66.5 months after completing RT, which far exceeds the median in group 2, and likely reflects a strong selection bias towards patients with good response to systemic therapy and stable metastatic disease.
Radiation skin toxicity observed in this study was much higher than rates reported with standard post-operative radiotherapy (3,10). Moist desquamation occurred in 60% of cases, likely due to the high proportion of patients with tumour skin invasion (79%) which would necessitate the use of bolus material to intentionally increase the radiation dose at the skin surface. Other factors which may have contributed to the high degree of skin toxicity include concurrent chemotherapy in six patients (14%), higher radiation dose, and the use of twice daily fractionation schedules. None of the patients in this study experienced grade 4 or 5 radiation dermatitis, and an improvement in symptoms such as bleeding and ulceration was recorded in 13 (54%) of applicable cases. One patient developed clinically significant radiation pneumonitis which resolved with corticosteroids.
Most patients in this study demonstrated tumour shrinkage and symptom relief in response to RT, and the maximal response was generally seen within 3 months of the last treatment. Many patients on this study were offered RT after failing to respond or progressing on systemic therapy. Thirty percent of patients in this study eventually developed locoregional progression; although the median LPFS was lower in group 2 (8.2 months) than group 1 (13.4 months), the difference between the two was statistically insignificant when evaluated using the Kaplan-Meier method and log-rank test. Our interpretation of this result is that it demonstrates a clinically significant benefit from RT in these patients who had very aggressive disease and limited treatment options remaining.
Reports of OS in patients with LABC range from 44–132 months (14,15), and approximately 20–35 months in patients with distant metastases at presentation (6,8). The median OS in group 1 (15.9 months) and group 2 (13.7 months) were lower than has been reported in the literature. This may be in part because previous studies calculated OS from the time of diagnosis, whereas in this study OS was measured from completion of RT. Interestingly, patients in group 1 did not have significantly better OS than patients in group 2, despite being treated at an earlier disease stage. Patients were generally younger in group 2 than in group 1, which may have contributed to their longer OS. Furthermore, it is possible that some patients in group 1 were given RT with very little likelihood of subsequent surgery due to tumour characteristics or medical comorbidities; this may have contributed to the low OS in group 1. The study’s small sample size also limits the utility of comparisons between the groups.
This study was retrospective in design, and was therefore subject to several limitations, including a heterogeneous patient population, variable follow-up, and the inability to assess tumour response using standardized criteria, such as RECIST. Radiation treatments also varied significantly, reflecting the lack of evidence to guide the use of RT for unresected disease. To our knowledge, seventeen patients in this cohort were alive at the time of this review and censored at their last follow-up at our centre. Longer follow-up would be helpful to determine the exact outcome of these patients; however, OS and LPFS were determined using the Kaplan-Meier method which is designed to account for patients who are lost to follow-up before an event is observed (16,17). The overall sample size was small considering the long inclusion period and the large number of breast cancer patients seen at the institution, which reflects this uncommon and clinically challenging scenario. Prospective studies of radiotherapy for unresected disease that utilize standard radiation regimens and response criteria as well as patient reported outcomes are needed to better characterize the potential benefits of treatment.
Conclusions
Most LABC patients in this study demonstrated at least a partial response to RT and a decrease in tumour-related symptoms where applicable. In most cases, the maximal RT response occurred within 3 months of completing treatment. Locoregional RT is an effective option for management of symptoms and tumour burden in patients with unresected, locally-advanced breast cancer.
Acknowledgements
Dr. J Lee is the senior author and Principal Investigator of this study. We thank the generous support of Bratty Family Fund, Michael and Karyn Goldstein Cancer Research Fund, Joey and Mary Furfari Cancer Research Fund, Pulenzas Cancer Research Fund, Joseph and Silvana Melara Cancer Research Fund, and Ofelia Cancer Research Fund.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the institutional ethics board of Sunnybrook Health Sciences Centre (No. 071-2014).
References
- Raphael J, Paramsothy T, Li N, et al. A single-institution experience of salvage therapy for patients with early and locally advanced breast cancer who progress during neoadjuvant chemotherapy. Breast Cancer Res Treat 2017;163:11-9. [Crossref] [PubMed]
- El Saghir NS, Eniu A, Carlson RW, et al. Locally advanced breast cancer: Treatment guideline implementation with particular attention to low- and middle-income countries. Cancer 2008;113:2315-24. [Crossref] [PubMed]
- Brackstone M, Palma D, Tuck AB, et al. Concurrent neoadjuvant chemotherapy and radiotherapy in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 2017;99:769-76. [Crossref] [PubMed]
- McGuire SE, Gonzalez-Angulo AM, Huang EH, et al. Postmastectomy radiation improves the outcome of patients with locally advanced breast cancer who achieve a pathologic complete response to neoadjuvant chemotherapy. Int J Radiat Oncol Biol Phys 2007;68:1004-9. [Crossref] [PubMed]
- Huang EH, Tucker SL, Strom EA, et al. Postmastectomy radiation improves local-regional control and survival for selected patients with locally advanced breast cancer treated with neoadjuvant chemotherapy and mastectomy. J Clin Oncol 2004;22:4691-9. [Crossref] [PubMed]
- Badwe R, Hawaldar R, Nair N, et al. Locoregional treatment versus no treatment of the primary tumour in metastatic breast cancer : an open-label randomised controlled trial. Lancet Oncol 2015;16:1380-8. [Crossref] [PubMed]
- Truong PT. Local treatment of the primary tumor in patients presenting with stage IV breast cancer: A first, and what’s up ahead. Int J Radiat Oncol Biol Phys 2017;97:443-6. [Crossref] [PubMed]
- Le Scodan R, Stevens D, Brain E, et al. Breast cancer with synchronous metastases: Survival impact of exclusive locoregional radiotherapy. J Clin Oncol 2009;27:1375-81. [Crossref] [PubMed]
- Moran MS. Radiation therapy in the locoregional treatment of triple-negative breast cancer. Lancet Oncol 2015;16:e113-22. [Crossref] [PubMed]
- Kao J, Conzen SD, Jaskowiak NT, et al. Concomitant radiation therapy and paclitaxel for unresectable locally advanced breast cancer: Results from two consecutive Phase I/II trials. Int J Radiat Oncol Biol Phys 2005;61:1045-53. [Crossref] [PubMed]
- Gaui MF, Amorim G, Arcuri RA, et al. A phase II study of second-line neoadjuvant chemotherapy with capecitabine and radiation therapy for anthracycline-resistant locally advanced breast cancer. Am J Clin Oncol 2007;30:78-81. [Crossref] [PubMed]
- Kosma L, Koukourakis M, Skarlatos J, et al. Hypofractionated radiotherapy with 5-fluorouracil radiosensitization for locally “far advanced” breast cancer. Am J Clin Oncol 1997;20:562-6. [Crossref] [PubMed]
- Bourgier C, Khodari W, Vataire AL, et al. Breast radiotherapy as part of loco-regional treatments in stage IV breast cancer patients with oligometastatic disease. Radiother Oncol 2010;96:199-203. [Crossref] [PubMed]
- Fried G, Semenisty V, Regev Z, et al. Patterns of care and outcome of locally advanced breast cancer. Eur J Cancer 2012;48:S112. [Crossref]
- Brito RA, Valero V, Buzdar AU, et al. Long-Term Results of Combined-Modality Therapy for Locally Advanced Breast Cancer with ipsilateral supraclavicular metastases: the University of Texas M.D. Anderson cancer centre Experience. J Clin Oncol 2001;19:628-33. [Crossref] [PubMed]
- Kaplan EL, Meier P. Nonparametric Estimation from Incomplete Observations. J Am Stat Assoc 1958;53:457-81. [Crossref]
- Rich JT, Neely JG, Paniello RC, et al. A practical guide to understanding Kaplan-Meier curves. Otolaryngol Head Neck Surg 2010;143:331-6. [Crossref] [PubMed]