A pilot study with palonosetron in the prophylaxis of radiation-induced nausea and vomiting
Introduction
A common disease- and treatment-related side effect, nausea and vomiting remains a prevalent and distressing issue for patients with cancer, despite improved understanding of prevention and treatment of emesis in the past two decades. The volume of research on radiation-induced nausea and vomiting (RINV) is dwarfed by that of its counterpart—chemotherapy-induced nausea and vomiting (CINV). This gap in knowledge and its translation into clinical practice is evident: in current antiemetic guidelines, with RINV recommendations lacking similar strength as those in CINV (1); in international patterns of practice, with poor risk estimation and non-adherence to guidelines worldwide (2); and in patient experience, with reports of insufficient antiemetic treatment and high incidence of RINV (3).
Several large observational studies in patients without anti-emetic treatment have reported incidences of RINV at approximately 30–40% overall, and 50–80% in those who receive abdominal radiation (3,4). It is postulated that, similar to the mechanism of CINV, radiation induces nausea and vomiting by damaging the gastrointestinal mucosa, releasing serotonin, activating 5-hydroxytryptamine 3 (5-HT3) receptors and sending a signal to the brainstem to produce the response (5). Left untreated, prolonged emesis can impede on physical and social functioning, with worrisome impact on appetite, nutrition, and weight (6-8). In the clinical context, emesis may lead to treatment delay or termination, or affect compliance with other therapies, compromising efficacy (9).
In an effort to elucidate appropriate treatment of RINV, the American Society of Clinical Oncology (ASCO), Multinational Association of Supportive Care in Cancer/European Society for Medical Oncology (MASCC/ESMO), and the National Comprehensive Cancer Network (NCCN) have compiled anti-emetic guidelines for this particular patient population, endorsing prophylactic and rescue regimens with various agents including 5-HT3 receptor antagonists (RA), dopamine RAs and dexamethasone (10-12). For treatment areas carrying moderate (60–90%) emetic risk (upper abdomen), MASCC recommends prophylaxis with a 5-HT3 RA and optional dexamethasone. For low (30–60%) emetic risk (head and neck, thorax, pelvis), they recommend prophylaxis or rescue with dexamethasone, a dopamine RA or 5-HT3. Similarly, the NCCN recommends two first-generation 5-HT3 RAs (ondansetron or granisetron), with or without dexamethasone (12). Other proposed predictive treatment factors (e.g., concomitant chemotherapy) and patient factors (e.g., age, sex, prior chemotherapy) of RINV are acknowledged in the guidelines, however not accounted for in risk estimation.
A second generation 5-HT3 RA, palonosetron (Aloxi®), is characterized by a higher selective binding affinity and a longer half-life of 40 hours, compared to its predecessors ondansetron and granisetron (13). Its efficacy and safety in the chemotherapy setting is well-established. A systematic review and meta-analysis conducted by Popovic et al. observed palonosetron to be statistically superior to first generation 5-HT3 RAs in several endpoints, including the percentage of patients achieving complete response (no emetic episode/use of rescue medication), complete control (no emetic episode/use of rescue medication and no more than mild nausea), no emesis, no nausea, and no use of rescue medication (14). Owing to its pharmacology, palonosetron is the only 5-HT3 RA at present to have a specific indication for the prevention of delayed CINV (15).
The efficacy and safety of palonosetron has yet to be studied in the radiation setting. The purpose of this pilot study was to evaluate control rates of emesis in patients without pre-existing nausea and vomiting treated with palliative radiotherapy and prophylactic palonosetron.
Methods
Patients and treatment
Patients were eligible if they were undergoing palliative radiotherapy to sites categorized as moderate or low risk for RINV by the cooperative guidelines from MASCC/ESMO. Only patients without nausea, vomiting, or anti-emetic use at baseline were included. The study was approved by the hospital research ethics board and Health Canada. All patients provided informed consent. Patients who had received cranial radiation or chemotherapy within 7 days preceding the start of radiotherapy, or were scheduled to receive such treatments during or within 10 days of study treatment were ineligible. Patients were not eligible if they were scheduled to receive corticosteroid treatment within 48 hours preceding, during or within 10 days following radiotherapy. Patients with poor performance status [Karnofsky Performance Status (KPS) <40] were excluded.
Patients received radiation with one of the three following dose/fractionation schedules: 8 Gray (Gy) in 1 fraction, 20 Gy in 5 fractions or 30 Gy in 10 fractions. Patients were pre-medicated with 0.5 mg of palonosetron orally, at least 1 hour prior to the first fraction of radiation treatment, and every other day until the completion of radiation treatment. Patients were to receive palonosetron on weekends and holidays within the scheduled treatment to ensure continuous coverage.
Using a two-sided binomial hypothesis test with a target significance level of 5%, a sample size of 60 would be required to detect a complete control of vomiting at 85% with palonosetron compared to a historical complete control of vomiting at 70% with ondansetron. This sample size achieved 80% power to detect the difference between 70–85%. With a 20% drop-out rate, it was calculated that 75 patients would be enrolled.
Data collection
Patient demographic and medical information were collected including age, sex, primary cancer site, performance status, previous and concurrent systemic therapies, and prescribed radiation treatment.
Patients completed a daily diary at baseline, every day during radiation treatment (including weekends and holidays) and for 10 days post-treatment. The daily diary assessed vomiting, nausea, diarrhea, the interference of any existing RINV in their daily life, as well as their use of anti-emetics. Nausea, vomiting and diarrhea were rated by the patient on level of severity (none, mild, moderate or severe). Patients who had nausea or vomiting were asked to record the number of episodes they experienced and rank its interference with aspects of daily life on a 5-point scale in daily diaries. Research assistants maintained copies of daily diaries through regular telephone follow-ups. Patient diaries were collected at the end of the study period.
Patient-reported adverse events were monitored as per Common Terminology Criteria for Adverse Events Version 4 guidelines and recorded at baseline as well as during follow-up calls with research assistants. Patients completed the validated Functional Life Index-Emesis (FLIE) and the European Organisation for Research and Treatment of Cancer Quality of Life (QOL) Questionnaire Core 15-Palliative (C15-PAL) at baseline and again at regular intervals during and post- treatment.
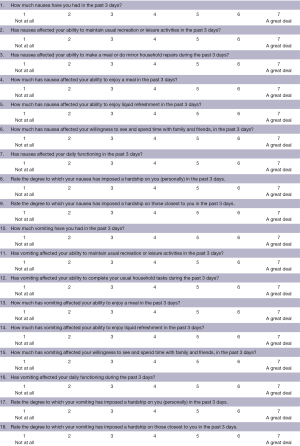
The FLIE evaluates the prevalence of nausea (Q1) and vomiting (Q10), as well as their impact on various aspects of function and QOL (nausea: Q2–9; vomiting: Q11–18) in a recall period of 3 days, on a 7-point scale (Figure S1) (16). Patients completed the FLIE at baseline, days 5 and 10 during treatment (for multiple fractions of radiotherapy), and days 3 and 7 following treatment.
The C15-PAL is a module specific to the palliative patient population, and is composed of 15 questions spread amongst 2 multi-item functional scales, 2 multi-item symptom scales, 5 individual symptom questions and 1 question on overall QOL (Figure S2) (17). All questions use a 4-point scale, with an exception of the overall QOL question, which is answered on a 7-point scale. Higher scores indicate greater functionality, better QOL, and worse symptomology on the respective scales. Patients completed the C15-PAL at baseline, days 5 and 10 during treatment (if applicable), and days 5 and 10 following treatment.
Study definitions
Patients were followed during acute (day 1 of treatment to day 1 post-treatment) and delayed (days 2–10 post-treatment) phases of treatment.
Nausea was defined as a feeling occurring in the areas of the back of the throat to the stomach, often described as queasiness. Nausea may or may not have led to vomiting. Vomiting was defined as the oral forceful expulsion of stomach contents. Other terms used were “throwing up” or “puking”. An episode of vomiting had a distinct starting and ending point, with at least one occurrence of vomiting in between. Individual episodes were separated by the lack of vomiting for at least 5 minutes. Side effects of constipation and headache were patient-reported and defined as per the individual.
Efficacy parameters
Anti-emetic clinical trials often use “complete response” as an endpoint denoting no emetic episodes or use of rescue medication (14). Study endpoints such as “complete control” and “partial control” used in our group’s previous anti-emetic trial were adopted (18). For the purpose of the present study, treatment response or “complete control” was defined using the same criteria. Partial control was defined as 1–2 episodes of nausea or vomiting, but no use of rescue medication. Three or more episodes of nausea or vomiting, or use of rescue medication was defined as uncontrolled response. Responders referred to patients with complete control.
Statistical analysis
Demographic and medical information were described in all patients as mean, standard deviation (SD), median, and range for continuous variables, and as proportions for categorical variables. Proportions of patients achieving complete control, partial control and uncontrolled response were calculated according to study definitions and presented separately for acute and delayed phases. FLIE and C15-PAL summary scores were also summarized at baseline, during treatment, and post treatment at day 5 and day 10, respectively. The proportion of patients with side effects (i.e., constipation, headache) was calculated at baseline, acute phase, and delayed phase. The level of severity and relation to treatment were also described for constipation and headache side effects.
To search for relationships, general linear regression models were performed at day 3 or at day 7 post-treatment. Mean squared error (MSE) was estimated for the average of the squares of the error (lower MSE, better fit). Natural log-transformation was applied for all FLIE items, except for Q5 to normalize the distribution.
Number of nausea or vomiting episodes was totaled for days 1–5 and days 6–10 post-treatment. Spearman correlations were calculated between total episodes of nausea/vomiting during these two periods and C15-PAL items at day 5 and day 10 respectively. The relationships between total episodes and C15-PAL items were conducted using generalized linear model for count data. The GENMOD procedure in Statistical Analysis Software (SAS) was performed, and Poisson distribution with log link function was used. To normalize the distribution, natural log-transformation was applied for all C15-PAL items. All analyses were conducted using SAS (version 9.4 for Windows). P value <0.05 was considered statistically significant.
Role of the funding source
The funding source had no role in the study design, data collection, analysis or interpretation. All authors had access to all data. The corresponding author had final responsibility for the decision to submit for publication.
Results
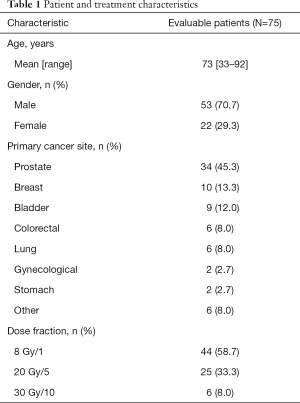
From April 2015 to July 2017, 75 patients without pre-existing nausea and vomiting at baseline were enrolled. All 75 patients were assessed in the acute phase. Due to two withdrawals where patients did not provide any information after the acute phase, there were 73 patients included in the delayed phase. Acute and delayed phase data was available for 75 and 73 patients, respectively. Patient and treatment characteristics are summarized in Table 1.
Full table
Efficacy endpoints
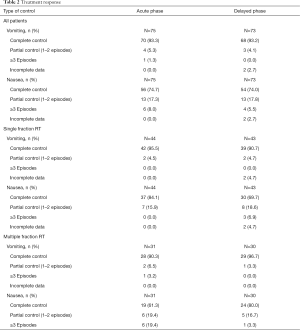
Treatment responses are summarized in Table 2. In the acute phase, 93.3% and 74.7% reported complete control of vomiting and nausea respectively. In the delayed phase, 93.2% and 74.0% reported complete control of vomiting and nausea respectively. Of the 75 patients, 2 non-responders reported use of rescue medication.
Full table
In patients who received a single fraction of radiation, 95.5% and 84.1% reported complete control of vomiting and nausea respectively in the acute phase (Table 2). In the delayed phase, rates of complete control of vomiting and nausea decreased to 90.7% and 69.7% respectively. In patients who received multiple fractions of radiation, 90.3% and 61.3% reported complete control of vomiting and nausea respectively in the acute phase (Table 2). In the delayed phase, rates of complete control of vomiting and nausea increased to 96.7% and 80.0% respectively.
Adverse events
At baseline, 3 (4.0%) and 27 (36.0%) patients reported headache and constipation, respectively. Side effects were evaluable in 71 patients, of which 9 (12.7%) and 50 (70.4%) reported headache and constipation respectively during the study period. All incidences of headache were mild. All reported cases of constipation were either mild (60.0%) or moderate (40.0%).
QOL
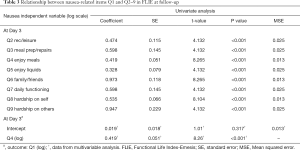
There were significant associations between Q1 (amount of nausea) of the FLIE and the remaining items related to nausea at both days 3 post-treatment (Table 3). In the multivariable analysis of nausea, only Q4 (enjoyment of food) remained significantly associated with Q1 at day 3. At day 7 follow-up, an analysis was not possible due to too few patients experiencing nausea at this time. A similar analysis was unable to be conducted using amount of vomiting due to low levels in the patient sample. Responders in delayed phase for nausea demonstrated lower nausea summary scores (Q1–Q9) at day 3 and 7 follow-up (Table 4).
Full table
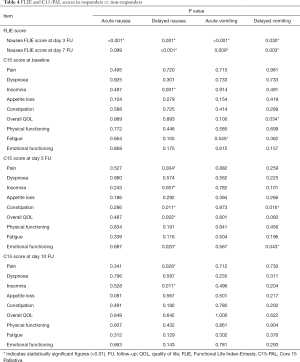
Full table
Responders in delayed phase for nausea had significantly lower C15-PAL scores in insomnia (P=0.007), pain (P=0.004), constipation (P=0.011), and emotional functioning (P=0.020) compared to non-responders at day 5 follow-up (Table 4). Responders in delayed phase for nausea also had significantly higher overall QOL (P=0.002). Pain (P=0.028) and insomnia (P=0.011) remained significantly lower in responders compared to non-responders at day 10 follow-up. Responders in delayed phase for vomiting had significantly lower C15-PAL scores in constipation (P=0.016) and emotional functioning (P=0.043) at day 5 follow-up (Table 4). There were no significant differences between responders and non-responders in delayed phase for vomiting at day 10 follow-up.
In a univariate analysis of C15-PAL scores at day 5 follow-up, there were significant associations between the number of episodes of nausea and both pain (P=0.021) and fatigue (P=0.033) (Table 5). Only pain remained significantly associated to number of nausea episodes in the multivariate analysis (P=0.021). Since there were no reports of nausea at day 10 follow-up, this analysis could only be conducted at day 5 of follow-up. A similar analysis between C15-PAL scores and number of episodes of vomiting did not yield significant results, most likely due to too few patients experiencing vomiting.

Full table
Discussion
To our knowledge, this is the first reported study using palonosetron in the prophylaxis of RINV. Our study suggests favorable control, as illustrated by rates of complete control of vomiting at approximately 93% in both acute and delayed phases. Similarly, complete control of nausea was approximately 74% in both phases.
In their randomized phase III study, Gralla et al. observed statistically higher rates of complete control (no emetic episodes/no rescue medicine and no more than mild nausea) in patients receiving moderately emetic chemotherapy who were prescribed 0.25 mg palonosetron compared to ondansetron in the acute (81.0% vs. 68.6%) and delayed (74.1% vs. 55.1%) phases, and in the study period overall (69.3% vs. 50.3%) (19). Current understanding of and guidance on future directions in RINV is derived from the chemotherapy setting. However, given the different mechanisms, timing and emetic risks of these treatments, it is difficult to make valid comparisons between the settings. Although it should be noted that a higher dose of palonosetron was prescribed, complete control of vomiting was superior in the present study compared to those observed by Gralla et al., at 93.3% and 93.2% in the acute and delayed phases respectively.
An analysis of treatment responses in a study published previously by our group using ondansetron in a similar patient population yielded complete control rates of 59% (nausea) and 75% (vomiting) in the acute phase, and 39% (nausea) and 52% (vomiting) in the delayed phase (20). Using the most comparable endpoint in the current study, complete control in nausea and vomiting with palonosetron exceeds complete control of ondansetron by 16% and 18% in the acute phase respectively, and by 35% and 41% in the delayed phase respectively. Another notable observation is the apparent maintenance of complete control from the acute to delayed phase with palonosetron, whereas complete control of nausea and vomiting fell by 20% and 23% with ondansetron. However, it should be noted that those who achieved control in the acute phase did not necessarily do so in the delayed phase, and vice versa. There was a decrease in complete control from acute to delayed phase vomiting in patients who received a single fraction of radiation, whereas there was an increase in complete control from acute to delayed phase vomiting in patients treated with multiple fractions of radiation. Patients were treated with radiation and the study medication during the acute phase. Those who received multiple fractions of radiation also consumed an increased amount of palonosetron compared to those who received a single fraction of radiation. The residual effect of these multiple doses may explain the differences in control in the delayed phase between these groups.
Results from an international survey of radiation oncologists indicated 5-HT3 RAs to be the antiemetic agent most commonly prescribed by clinicians (2). A systematic review and meta-analysis conducted by Li et al. demonstrated a significantly higher efficacy of 5-HT3 RAs (ondansetron, granisetron, dolasetron) compared to dopamine RAs in both complete control of vomiting (OR 0.17, 95% CI: 0.05–0.58) and nausea (OR 0.46, 95% CI: 0.24–0.88) (21).
There are some limitations of the study results. These limitations also applied to our previous study employing ondansetron. Patients received moderate or low risk radiation treatment. As such, these results may not be generalizable to those receiving high risk treatment. Patients were mostly geriatric, suffered from painful bone metastases, and were on several medications including opioids for cancer-related pain, which also pose a risk of nausea and constipation.
Although results from the current study suggest improved control with palonosetron versus ondansetron, a randomized controlled phase III trial is needed to assess superiority. Given that more than half of those with cancer will receive radiation during the course of their disease, there is great potential for improved well-being with research in RINV (22).
Conclusions
There is suggested improved prophylaxis of treatment-induced nausea and vomiting with palonosetron in the radiation setting. It was safe and well-tolerated by study patients. Efficacy of palonosetron compared to previous 5-HT3 RAs should be investigated with a randomized trial.
Acknowledgements
We thank Canadian Eisai for the supply of the medication.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the hospital research ethics board and Health Canada (ID: 434-2013). All patients provided informed consent.
References
- Dennis K, Maranzano E, De Angelis C, et al. Radiotherapy-induced nausea and vomiting. Expert Rev Pharmacoecon Outcomes Res 2011;11:685-92. [Crossref] [PubMed]
- Dennis K, Zhang L, Lutz S, et al. International patterns of practice in the management of radiation therapy-induced nausea and vomiting. Int J Radiat Oncol Biol Phys 2012;84:e49-60. [Crossref] [PubMed]
- Enblom A, Bergius Axelsson B, Steineck G, et al. One third of patients with radiotherapy-induced nausea consider their antiemetic treatment insufficient. Support Care Cancer 2009;17:23-32. [Crossref] [PubMed]
- Maranzano E, De Angelis V, Pergolizzi S, et al. A prospective observational trial on emesis in radiotherapy: analysis of 1020 patients recruited in 45 Italian radiation oncology centres. Radiother Oncol 2010;94:36-41. [Crossref] [PubMed]
- Minami M, Endo T, Hirafuji M, et al. Pharmacological aspects of anticancer drug-induced emesis with emphasis on serotonin release and vagal nerve activity. Pharmacol Ther 2003;99:149-65. [Crossref] [PubMed]
- Nguyen J, Leung M, Presutti R, et al. Position of appetite and nausea in symptom clusters in palliative radiation therapy. Preedy V, editor. Taylor & Francis Group, 2011.
- Collis E, Mather H. Nausea and vomiting in palliative care. BMJ 2015;351:h6249. [Crossref] [PubMed]
- Poon M, Hwang J, Dennis K, et al. A novel prospective descriptive analysis of nausea and vomiting among patients receiving gastrointestinal radiation therapy. Support Care Cancer 2016;24:1545-61. [Crossref] [PubMed]
- Ballatori E, Roila F. Impact of nausea and vomiting on quality of life in cancer patients during chemotherapy. Health Qual Life Outcomes 2003;1:46. [Crossref] [PubMed]
- Basch E, Prestrud AA, Hesketh PJ, et al. Antiemetics: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 2011;29:4189-98. Erratum in: J Clin Oncol 2014;32:2117. [Crossref] [PubMed]
- Roila F, Molassiotis A, Herrstedt J, et al. 2016 MASCC and ESMO guideline update for the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting and of nausea and vomiting in advanced cancer patients. Ann Oncol 2016;27:v119-v33. [Crossref] [PubMed]
- Network NCC. Antiemesis. 2017. Available online: https://www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf
- Aapro MS. Palonosetron as an anti-emetic and anti-nausea agent in oncology. Ther Clin Risk Manag 2007;3:1009-20. [PubMed]
- Popovic M, Warr DG, Deangelis C, et al. Efficacy and safety of palonosetron for the prophylaxis of chemotherapy-induced nausea and vomiting (CINV): a systematic review and meta-analysis of randomized controlled trials. Support Care Cancer 2014;22:1685-97. [Crossref] [PubMed]
- Celio L, Niger M, Ricchini F, et al. Palonosetron in the prevention of chemotherapy-induced nausea and vomiting: an evidence-based review of safety, efficacy, and place in therapy. Core Evid 2015;10:75-87. [Crossref] [PubMed]
- Lindley CM, Hirsch JD, O'Neill CV, et al. Quality of life consequences of chemotherapy-induced emesis. Qual Life Res 1992;1:331-40. [Crossref] [PubMed]
- Groenvold M, Petersen MA, Aaronson NK, et al. The development of the EORTC QLQ-C15-PAL: a shortened questionnaire for cancer patients in palliative care. Eur J Cancer 2006;42:55-64. [Crossref] [PubMed]
- Wong E, Pulenzas N, Bedard G, et al. Ondansetron rapidly dissolving film for the prophylactic treatment of radiation-induced nausea and vomiting-a pilot study. Curr Oncol 2015;22:199-210. [Crossref] [PubMed]
- Gralla R, Lichinitser M, Van Der Vegt S, et al. Palonosetron improves prevention of chemotherapy-induced nausea and vomiting following moderately emetogenic chemotherapy: results of a double-blind randomized phase III trial comparing single doses of palonosetron with ondansetron. Ann Oncol 2003;14:1570-7. [Crossref] [PubMed]
- Dennis K, Nguyen J, Presutti R, et al. Prophylaxis of radiotherapy-induced nausea and vomiting in the palliative treatment of bone metastases. Support Care Cancer 2012;20:1673-8. [Crossref] [PubMed]
- Li WS, van der Velden JM, Ganesh V, et al. Prophylaxis of radiation-induced nausea and vomiting: a systematic review and meta-analysis of randomized controlled trials. Ann Palliat Med 2017;6:104-17. [Crossref] [PubMed]
- Institute NC. Radiation therapy for cancer. 2010; Available online: https://www.cancer.gov/about-cancer/treatment/types/radiation-therapy/radiation-fact-sheet