Palliative radiotherapy utilization within a regional Australian palliative care unit
Introduction
Palliative care aims to improve the quality of life (QOL) of patients with both malignant and non-malignant diseases. For patients with cancer, increasingly this means earlier integration into oncological management through multi-disciplinary team based approaches. Concurrent involvement of oncological and palliative care services has been demonstrated to improve symptom control and enhance QOL (1). The importance of the dual involvement of radiation oncology and palliative care is increasingly recognized given both specialties are commonly involved with patients with advanced malignant disease (2).
Palliative radiotherapy has been demonstrated to be cost-effective and efficacious for managing a range of symptoms in both locally advanced and metastatic cancers (3,4). Common indications for use include pain from metastatic disease, neurological dysfunction secondary to brain or spinal disease, malignant bleeding and obstructive symptoms. Traditionally palliative radiotherapy has been shorter in duration with increased consideration of cost, convenience and avoidance of adverse effects than curative radiotherapy (2,3,5).
While there is support for the efficacy of palliative radiotherapy across a range of indications, concerns have been raised about its appropriateness as patients deteriorate from advanced disease (2,5-8). Factors requiring particular consideration in this setting include the inherent inaccuracy of prognostic prediction (2), variable latency to therapeutic effect (6), side effects and the logistics for the patient, families and health care providers of getting to treatment. Rates of radiotherapy use at end of life vary (8,9) and there is no established optimal rate (5). Both benefit and harm have been demonstrated for palliative radiotherapy in the last months of life (6,9,10) although variance exists depending upon indication for use (5). The use of chemotherapy in the last 14 days of life has been considered an indicator of poorer care (5), and some have suggested that radiotherapy provision in the last 14 or 30 days of life might also be a useful quality marker (7).
Given many symptom and clinical issues arising in advanced malignancy represent a nexus between radiation oncology and inpatient palliative care, it is perhaps surprising that published literature specifically investigating this interface is limited. In one of the few published papers addressing this question Al-Shahri et al. looked at referrals to radiation oncology from a Saudi Arabian palliative care unit (PCU) (11). Four percent of 635 cancer admissions across approximately 40 months were referred with pain control the commonest reason for referral (88% of referrals). The median survival time post radiotherapy in this study was 30 days. A poster at the recent European Association of Palliative Care Congress, investigated outcomes for PCU inpatients receiving radiotherapy within an Australian metropolitan health service (12). In this retrospective audit of 119 patients more than 40% of patients died during their admission, with 29% and 45% dying within 14 and 30 days of radiotherapy completion respectively. Given the lack of data, vulnerability of this patient group and potential for both benefit and harm, the question of radiation oncology utilization amongst patients in our local PCU was raised.
The aims of the current project were to evaluate the utilization of palliative radiotherapy amongst inpatient palliative care patients including evaluation of how, why, where, when and to whom palliative radiotherapy was provided. It was hoped these data would clarify current service utilization as well as provide insights into the outcomes of palliative radiotherapy in the PCU, which might then be used to guide future clinical practice.
Methods
A retrospective cohort study was undertaken utilising routinely collected data. All patients admitted to the Barwon Health PCU between August 2011 and June 2016 who underwent palliative radiotherapy whilst an inpatient were included. This cohort included those who were receiving radiotherapy at the time of admission and those who commenced radiotherapy during their admission. Of note the PCU is on a separate campus approximately five kilometers from the acute hospital where radiotherapy takes place. While the uptake of radiotherapy by Barwon Health PCU inpatients was not able to be specified a priori, it was felt that 5 years of data would provide an adequately representative sample. Data were extracted from Barwon Health palliative electronic medical record (PERM), Barwon Health digital medical records and Barwon Health radiation oncology databases. The project was approved by the Barwon Health Research Ethics, Governance & Integrity Unit (Barwon Health reference 16/190).
Variables collected
Demographic data collected included age, gender, place of residence prior to PCU admission and primary language spoken. Clinical variables included primary cancer diagnosis, reason for admission, palliative care phase on admission and performance status on admission. Radiotherapy-specific data analyzed included primary reason for radiotherapy, number of fractions, anatomical sites of treatments, whether treatment was completed and whether concurrent corticosteroids were prescribed.
In an attempt to assess benefit, opioid consumption was recorded (where available) for those patients who received radiotherapy specifically for pain. Mean total opioid use was calculated for the week prior to radiotherapy and then for each subsequent week post-radiotherapy. Total opioid usage was converted to oral morphine equivalent (OME) doses using an accepted opioid dose conversion table (13). Outcomes of admission were recorded and the time between commencement of radiotherapy and death calculated.
Analysis
Demographic data were analyzed descriptively. Frequency counts and percentages were used to summarize categorical variables, and mean (standard deviation) and/or median (interquartile range) for continuous variables. For episodes during which palliative radiotherapy was used specifically for pain control, non-parametric Wilcoxon Signed Rank Tests were used to compare opioid consumption before and after radiotherapy at time points 1, 2 and 3 weeks. Only episodes that had complete opioid consumptions data sets for each of the time points were included. Level of significance was set at <0.05. Survival following commencement of radiotherapy was analyzed using Kaplan-Meier curve. Analysis was undertaken using SPSS V.24 (IBM SPSS Statistics for Windows, Version 24.0. Armonk, NY: IBM Corp).
Results
Sixty episodes or courses of radiotherapy were provided to 51 PCU patients during the study period. Six patients had two separate courses of radiotherapy during the same admission with only the first course included in the analysis to maintain independence between subjects. This meant a total of 54 PCU admissions were included in the final analysis. This equated to approximately three percent of total PCU admissions (malignant and non-malignant) during the study period.
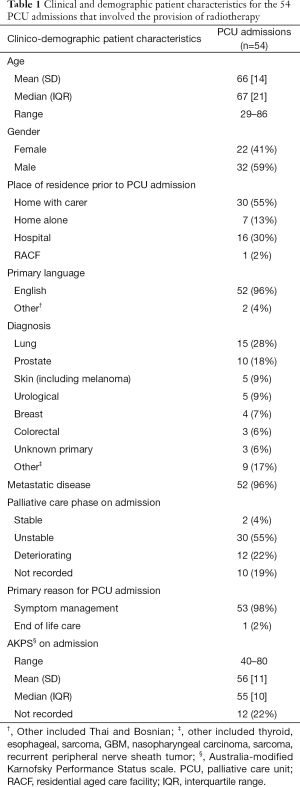
As shown in Table 1, the majority of patients were male and residing at home prior to PCU admission. Lung and prostate cancer were the commonest malignancies and virtually all patients had metastatic disease. In keeping with advanced disease more than 75% of patients were either clinically unstable or deteriorating at admission. Performance status varied however the average Australia-modified Karnofsky Performance Status (AKPS) score was between 50 and 60 representing the need for occasional to moderate assistance with care needs (14).
Full table
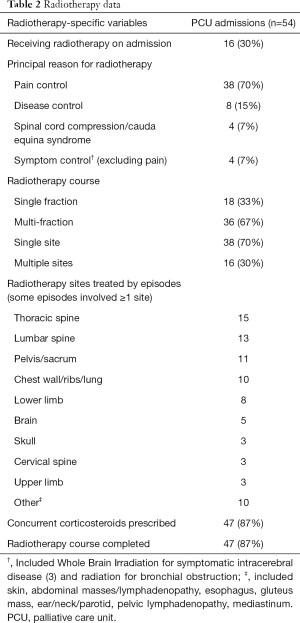
Most radiotherapy episodes were commenced once patients were established inpatients, with only 30% ongoing at the time of admission (Table 2). Pain management was the commonest reason for radiotherapy treatment and most courses were multi-fractionated. Approximately a third of episodes involved the treatment of more than site, with the thoracic and lumbar spines and pelvis the sites most commonly irradiated (either alone or in combination with other regions). Almost 90% of radiotherapy courses were completed and concurrent corticosteroids very frequently prescribed.
Full table
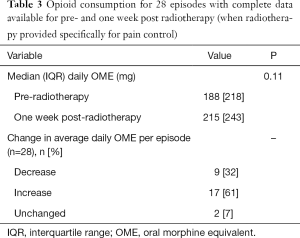
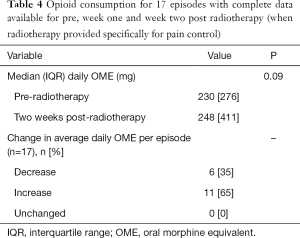
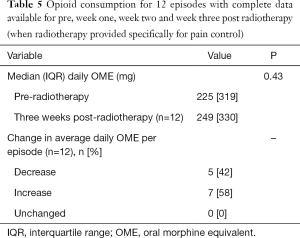
Tables 3-5, show changes in opioid consumption between baseline (week pre-radiotherapy) and each of week one, two and three post radiotherapy. At each of the time points there were proportionally more episodes in which opioid consumption increased than decreased although the proportion in which a decrease occurred improved each week. There was a trend towards an increase in median daily OME at each of the time points; however this did not reach statistical significance.
Full table
Full table
Full table
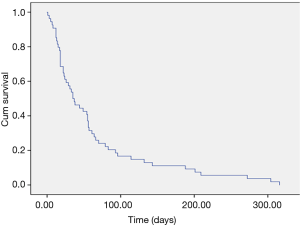
The commonest discharge outcome for patients receiving radiotherapy while in PCU was death and the median time between commencement of radiotherapy and death was 36 days (Table 6 and Figure 1). Twenty and 43 percent of patients died within 14 and 30 days of radiotherapy commencement respectively. Due to high levels of missing data it was not possible to analyze the performance status of patients discharged alive from the PCU.

Full table
Discussion
This paper adds to the small but growing body of literature looking at the provision of radiotherapy to palliative care inpatients. This exploration is important due to the potential for radiotherapy to have both benefits and harms and the inherent vulnerability of many patients requiring PCU admission. It is interesting that this is the second recent project that has addressed this broad question suggesting that interest in this important issue is increasing (12).
The management of cancer-induced bone pain (CIBP) is a common referral reason for palliative radiotherapy as well as a frequent indication for PCU admission. This was supported by the current data where the commonest reason for radiotherapy was pain control and the commonest regions irradiated the axial skeleton and pelvis. While efficacy has been demonstrated for palliative radiotherapy in the management of CIBP (8,9) the clinical benefits in malignant epidural spinal cord compression and intracerebral metastatic disease at end of life are less clear (5). This was not able to be specifically addressed in the current study owing to the limited number of patients whose primary indication for radiotherapy was not pain control. This does however represent an area for future research and might be best assessed by the creation of large, prospective multi-centre database.
Changes in opioid consumption for PCU patients before and after radiotherapy were used as a surrogate for treatment efficacy in this study. To the best of our knowledge this has not been done before in this setting. Attempts were made to concurrently analyse pain scores as a further marker of response however this was not possible due to considerable and somewhat surprising inconsistencies in pain recording approaches within the PCU. Response rates of 60–70% for palliative radiotherapy in CIBP have been reported (3,4,6), however these rates were not replicated in the current study. The proportional of patients requiring an increase in opioid dosage outnumbered those whose opioid dose was reduced and there was an overall trend towards higher median daily OME doses for each of the assessed time points. Using the proportion of patients whose opioid dose decreased following radiotherapy as a marker for response, response rates in the current study ranged from 32–41%. This is more in keeping with the Dutch Bone Metastasis Study that reported a 45% response rate to radiotherapy amongst a subset of patients who survived ≤12 weeks (10).
There are a multitude of reasons for these findings including the diminishing dataset for each of the weeks post radiotherapy. This was unavoidable as some patients were receiving radiotherapy prior to PCU admission, while others were discharged or died before the three week post-radiotherapy time point. It is conceivable that post-radiotherapy pain flare (15) might have been a contributory factor to the low proportion of patients with decreased opioids and high proportion with increased opioids at the end of the first week following radiotherapy. Additionally the provision of opioid medications for unstable or deteriorating patients with advanced malignant disease within a PCU setting is commonly for reasons other than pain or may have been for pain unrelated to the site or sites of radiotherapy. Both these factors limit the applicability of using changes in opioid consumption as an indicator of radiotherapy efficacy and accordingly these results, while important, should be interpreted cautiously and within context.
The 14 and 30 days mortalities of 20% and 43% in the current study align with Kernick and colleagues who reported mortality rates of 29% and 45% for PCU patients receiving radiotherapy within a metropolitan Australian setting (12). While the median survival of 36 days corresponds to that from a Saudi Arabian PCU (11), it is four times longer than the median survival time for all patients who die in the Barwon Health PCU. Additionally while 48% of discharge outcomes in the study were death, this is less than the average Barwon Health PCU discharge to death rate of 65%. While it is difficult to make definitive judgements as to the relative benefit of radiotherapy for the population in the current study, these broad metrics when considered together might suggest that the PCU patients who received radiotherapy did not have substantially poorer outcomes when compared to other PCU patients.
The appropriateness of and indications for radiotherapy in palliative care inpatients remain undefined and influenced by a range of patient, clinician and health service factors. When radiotherapy is provided for pain control there is potential for improvements in QOL through direct analgesic benefit and the commensurate sparing of adverse-effect inducing opioid medications. However this potential for upside needs to be balanced against factors including difficulties in prognostication, latency of benefit, risk of adverse effect and physical burden associated with the treatment itself. Despite data demonstrating the efficacy equivalence of single and multi-fractionated radiotherapy regimens for the management of bony metastases (16), multi-fractionated courses are still commonly employed world-wide (5). In the current study when radiotherapy provided specifically for pain control was considered, single fractions were used 68% of the time. This compares very favourably with retrospective US data that found that only 3.3% of 3,050 patients receiving radiotherapy for metastatic prostate cancer were treated with a single fraction (17). The benefits of a single fraction include improved patient and care-giver convenience (16) and although the risk of retreatment is greater when compared to a multi-fractionated course this is unlikely to be relevant for many PCU patients. Given this it would seem reasonable that attempts be made to optimise the use of single fraction treatments when PCU patients with CIBP are being considered for radiotherapy.
There are a number of limitations with this study including the small sample size. The retrospective nature meant that only pre-collected data were available for analysis and this was particularly limiting when it came to pain reporting. Marked inconsistency in pain assessment approaches within the PCU became apparent and this meant that the use of pain scores as a surrogate for radiotherapy efficacy was not possible. Importantly however this has provided impetus for re-evaluation of objective symptom assessment methods within the organization. The project was undertaken at a single regional Australian PCU where radiotherapy was provided off-site and this impacts generalizability.
Despite these limitations this paper provides an overview of radiotherapy use within an inpatient palliative care setting and raises a number of important considerations in relation to the appropriate stratification of PCU inpatients to radiotherapy. Importantly if patient care and health service utilization is to be optimised, more data are required ideally from large, multi-centre prospective studies that incorporate symptom assessment, quality-of-life measures, function, performance scales and health economic analysis.
Acknowledgements
The authors would like to thank Michelle Shields for her invaluable assistance with data collection.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The project was approved by the Barwon Health Research Ethics, Governance & Integrity Unit (Barwon Health reference 16/190).
References
- Temel JS, Grier JA, Muzikansky A, et al. Early palliative care for patients with metastatic non-small-cell lung cancer. N Engl J Med 2010;363:733-42. [Crossref] [PubMed]
- Lutz ST, Jones J, Chow E. Role of radiation therapy in palliative care of the patient with cancer. J Clin Oncol 2014;32:2913-9. [Crossref] [PubMed]
- Sharma S, Hertan L, Jones J. Palliative radiotherapy: current status and future directions. Semin Oncol 2014;41:751-63. [Crossref] [PubMed]
- Chow E, Harris K, Fan G, et al. Palliative radiotherapy trials for bone metastases: a systematic review. J Clin Oncol 2007;25:1423-36. [Crossref] [PubMed]
- Jones JA, Lutz ST, Chow E, et al. Palliative radiotherapy at the end of life: a critical review. CA Cancer J Clin 2014;64:296-310. [Crossref] [PubMed]
- Dennis K, Wong K, Zhang L, et al. Palliative radiotherapy for bone metastases in the last 3 months of life: worthwhile or futile? Clin Oncol (R Coll Radiol) 2011;23:709-15. [Crossref] [PubMed]
- Rautakorpi LK, Mäkelä JM, Seyednasrollah F, et al. Assessing the utilization of radiotherapy near end of life at a Finnish University Hospital: a retrospective cohort study. Acta Oncol. 2017;56:1265-71. [Crossref] [PubMed]
- Anshushaug M, Gynnild MA, Kaasa S, et al. Characterization of patients receiving palliative chemo-and radiotherapy during end of life at a regional cancer center in Norway. Acta Oncol 2015;54:395-402. [Crossref] [PubMed]
- Tiwana MS, Barnes M, Kiraly A, et al. Utilization of palliative radiotherapy for bone metastases near end of life in a population-based cohort. BMC Palliat Care 2016;15:2. [Crossref] [PubMed]
- Meeuse JJ, van der Linden YM, van Tienhoven G, et al. Efficacy of radiotherapy for painful bone metastases during the last 12 weeks of life. Cancer 2010;116:2716-25. [PubMed]
- Al-Shahri MZ, Al-Omair A, Al-Shabanah M, et al. Utilization of radiotherapy services by a palliative care unit: pattern and implication. J Support Oncol 2013;11:82-5. [PubMed]
- Kernick L, Moran J, Khor R. Outcomes of patients receiving inpatient radiotherapy on the palliative care unit. In: EAPC 15th World Congress of the European Association of Palliative Care Progressing Palliative Care Abstracts. Hayward Medical Communications, Newmarket, 2017;279.
- Australian and New Zealand College of Anaesthetists Faculty of Pain Medicine. Opioid Dose Equivalence [Internet]. Melbourne(AU);2016. Available online: http://fpm.anzca.edu.au/documents/opioid-dose-equivalence
- Abernethy AP, Shelby-James T, Fazekas BS, et al. The Australia-modified Karnofsky Performance Status (AKPS) scale: a revised scale for contemporary palliative care clinical practice BMC Palliat Care 2005;4:7. [ISRCTN81117481]. [Crossref] [PubMed]
- Gomez-Iturriaga A, Cacicedo J, Navarro A, et al. Incidence of pain flare following palliative radiotherapy for symptomatic bone metastases: multicenter prospective observational study. BMC Palliat Care 2015;14:48. [Crossref] [PubMed]
- Lutz S, Berk L, Chang E, et al. Palliative radiotherapy for bone metastases: an ASTRO evidence-based guideline. Int J Radiat Oncol Biol Phys 2011;79:965-76. [Crossref] [PubMed]
- Bekelman JE, Epstein AJ, Emanuel EJ. Single-vs multiple-fraction radiotherapy for bone metastases from prostate cancer. JAMA 2013;310:1501-2. [Crossref] [PubMed]